Advertisements
Advertisements
Describe the general trends in the following properties of the elements in Groups 13 and 14.
Metallic character
Concept: undefined >> undefined
Describe the general trends in the following properties of the elements in Groups 13 and 14.
Oxidation states
Concept: undefined >> undefined
Advertisements
Describe the general trends in the following properties of the elements in Groups 13 and 14.
Nature of halides
Concept: undefined >> undefined
Account for the following observations:
Though fluorine is more electronegative than chlorine yet BF3 is a weaker Lewis acid than BCl3
Concept: undefined >> undefined
Account for the following observations:
PbO2 is a stronger oxidising agent than SnO2
Concept: undefined >> undefined
Account for the following observations:
The +1 oxidation state of thallium is more stable than its +3 state.
Concept: undefined >> undefined
Three pairs of compounds are given below. Identify that compound in each of the pairs which has group 13 element in more stable oxidation state. Give reason for your choice. State the nature of bonding also.
TlCl3, TlCl
Concept: undefined >> undefined
Three pairs of compounds are given below. Identify that compound in each of the pairs which has group 13 element in more stable oxidation state. Give reason for your choice. State the nature of bonding also.
AlCl3 , AlCl
Concept: undefined >> undefined
Three pairs of compounds are given below. Identify that compound in each of the pairs which has group 13 element in more stable oxidation state. Give reason for your choice. State the nature of bonding also.
InCl3, InCl
Concept: undefined >> undefined
BCl3 exists as monomer whereas AlCl3 is dimerised through halogen bridging. Give reason. Explain the structure of the dimer of AlCl3 also.
Concept: undefined >> undefined
Boron fluoride exists as BF3 but boron hydride doesn’t exist as BH3. Give reason. In which form does it exist? Explain its structure.
Concept: undefined >> undefined
A nonmetallic element of group 13, used in making bullet proof vests is extremely hard solid of black colour. It can exist in many allotropic forms and has unusually high melting point. Its trifluoride acts as Lewis acid towards ammonia. The element exihibits maximum covalency of four. Identify the element and write the reaction of its trifluoride with ammonia. Explain why does the trifluoride act as a Lewis acid.
Concept: undefined >> undefined
Name the compounds whose line formulae are given below:

Concept: undefined >> undefined
Name the compounds whose line formulae are given below:
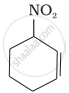
Concept: undefined >> undefined
Write structural formulae for compounds named as-
1-Bromoheptane
Concept: undefined >> undefined
Write structural formulae for compounds named as-
5-Bromoheptanoic acid
Concept: undefined >> undefined
Arrange the following in decreasing order of their boiling points.
(A) n-butane
(B) 2-methylbutane
(C) n-pentane
(D) 2, 2-dimethylpropane
Concept: undefined >> undefined
Match the reagent from Column I which on reaction with CH3 – CH = CH2 gives some product given in Column II as per the codes given below:
| Column I | Column II |
| (i) O3/Zn + H2O | (a) Acetic acid and CO2 |
| (ii) KMnO4/H+ | (b) Propan-1-ol |
| (iii) KMnO4/OH– | (c) Propan-2-ol |
| (iv) H2O/H+ | (d) Acetaldehyde and formaldehyde |
| (v) B2H6/NaOH and H2O2 | (e) Propane-1, 2-diol |
Concept: undefined >> undefined
Assertion (A): Among isomeric pentanes, 2, 2-dimethylpentane has highest boiling point.
Reason (R): Branching does not affect the boiling point.
Concept: undefined >> undefined
Which of the following gases is not a green house gas?
Concept: undefined >> undefined
