Please select a subject first
Advertisements
Advertisements
Name the part of the electromagnetic spectrum which is:
Suitable for radar systems used in aircraft navigation.
Concept: Electromagnetic Spectrum
Name the part of the electromagnetic spectrum which is:
Produced by bombarding a metal target with high electrons.
Concept: Electromagnetic Spectrum
Name the electromagnetic radiation whose frequency is 10 Hz.
Concept: Electromagnetic Spectrum
What is the speed of radio waves in vacuum?
Concept: Electromagnetic Spectrum
Name the electromagnetic radiation that has been used in obtaining the image below.
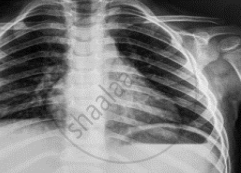
Concept: Electromagnetic Spectrum
What is the wavelength range of electromagnetic radiation used in radio broadcast?
Concept: Electromagnetic Spectrum
In an atom X, electrons absorb the energy from an external source. This energy “excites” the electrons from a lower-energy level to a higher-energy level around the nucleus of the atom. When electrons return to the ground state, they emit photons.
The figure below is the energy level diagram of atom X with three energy levels, E1 = 0.00eV, E2 = 1.78eV and E3 = 2.95eV. The ground state is considered 0 eV for reference. The transition of electrons takes place between levels E1 and E2.

- What wavelength of radiation is needed to excite the atom to energy level E2 from E1?
- Suppose the external source has a power of 100 W. What would be the rate of photon emission?
Concept: Electromagnetic Spectrum
Define critical angle for a given medium.
Concept: Total Internal Reflection
Name any two phenomena which take place in the formation of a rainbow.
Concept: Total Internal Reflection
The focal length of a double convex lens is equal to the radius of curvature of either surface. What is the refractive index of its material?
Concept: Power of a Lens
