Definitions [3]
Alkanes are hydrocarbons in which all the linkages between the carbon atoms are single covalent bonds.
Alkenes are unsaturated hydrocarbons containing at least one C=C double bond.
Alkynes are aliphatic unsaturated hydrocarbons containing at least one C≡C triple bond.
Key Points
- General formula: CₙH₂ₙ₊₂ (where n = number of carbon atoms)
- Suffix used for IUPAC naming: –ane
- e.g., Methane (CH₄), Ethane (C₂H₆), Propane (C₃H₈)
- Alkanes exhibit chain isomerism due to absence of any functional group and the possibility of more than one chain type for the same molecular formula
- e.g., C₅H₁₂ forms n-pentane, neo-pentane, and iso-pentane
Methods of Preparation:
| Method | Reactants → Conditions → Product |
|---|---|
| Catalytic hydrogenation of alkene | \[\ce{\underset{Alkene}{C_{n}H_{2n}} + H2 ->[\underset{(room temp)}{Pt or Pd}][\underset{(high temp and high pressure)}{or Ni}] C_{n}H_{2n} + 2}\] |
| Catalytic hydrogenation of alkyne | \[\ce{\underset{Alkene}{C_{n}H_{2n - 2}} + 2H2 ->[\underset{(room temp)}{Pt or Pd}][\underset{(high temp and high pressure)}{or Ni}] C_{n}H_{2n} + 2}\] |
| Reduction of alkyl halide | \[\ce{CH3—I + \underset{Nascent}{2[H]} ->[Zn, HCI] CH4 + HI}\] |
| Wurtz Coupling reaction | \[\ce{CH3 — Br + 2Na + CH3 — Br ->[Dry ether] CH3 — CH3 + 2NaBr}\] |
| From Grignard reagent |
\[\ce{CH3 — Mg — I + H2O ->[Dry ether] CH4 + MgI(OH)}\] |
| Soda lime decarboxylation |
\[\ce{RCOONa + NaOH ->[CaO/Δ] RH + Na2CO3}\] |
| Kolbe's electrolysis |
\[\ce{2RCOONa + H2O ->[Electrolysis] R — R + 2CO2 + 2NaOH + H2}\] |
1. Polarity:
- Alkanes are non-polar
- Insoluble in polar solvents (water)
- Soluble in non-polar solvents
2. Boiling point:
Increases with molecular mass
State at SATP:
- C₁–C₄ → gases
- C₅–C₁₇ → liquids
- C₁₈+ → solids
Boiling point order (increasing):
- Neo-pentane < iso-pentane < n-pentane
Trend:
- Straight-chain > branched-chain
- More branching → lower BP
3. Melting point:
- Increases with molecular mass
- Symmetrical molecules → higher MP
- Even number of C atoms → higher MP than odd
- Only intermolecular London forces are present
Alkanes are saturated and relatively inert — they undergo only substitution reactions at C–H bonds.
| Reaction | Conditions |
|---|---|
| Halogenation |
\[\ce{CH4 + Cl2 ->[hv][-HCl] CH3Cl ->[hv][-HCl] CH2Cl2 ->[hv][-HCl] CHCl3 ->[hv][-HCl] CCl4}\] |
| Combustion |
\[\ce{C_{n}H_{2n + 2} + \left(\frac{3n + 1}{2}\right)O_{2} ->[Complete][combustion] nCO2 + (n + 1)H2O}\] \[\ce{CH4(g) + 20_{2}(g) ->[Complete][combustion] CO2 + 2H2O}\] |
| Reforming / Aromatization |
\[\ce{n-hexane ->[V2O5][\underset{12-20 atm}{773K}] C6H6 + 4H2}\] |
| Pyrolysis / Cracking | 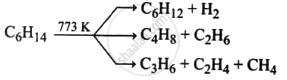 |
- The first four alkanes (CH₄, C₂H₆, C₃H₈, C₄H₁₀) are used as fuels (CNG, LPG).
- Lower liquid alkanes used as solvents.
- Used in making wax, ink, shoe polish, etc.
- Alkanes with more than 35 carbon atoms are used for road surfacing (tar).
- General formula: CₙH₂ₙ (where n = 2, 3, 4…)
- Suffix for IUPAC naming: –ene
- e.g., Ethene (CH₂=CH₂), Propene (CH₃–CH=CH₂)
- The double bond consists of one σ bond and one π bond
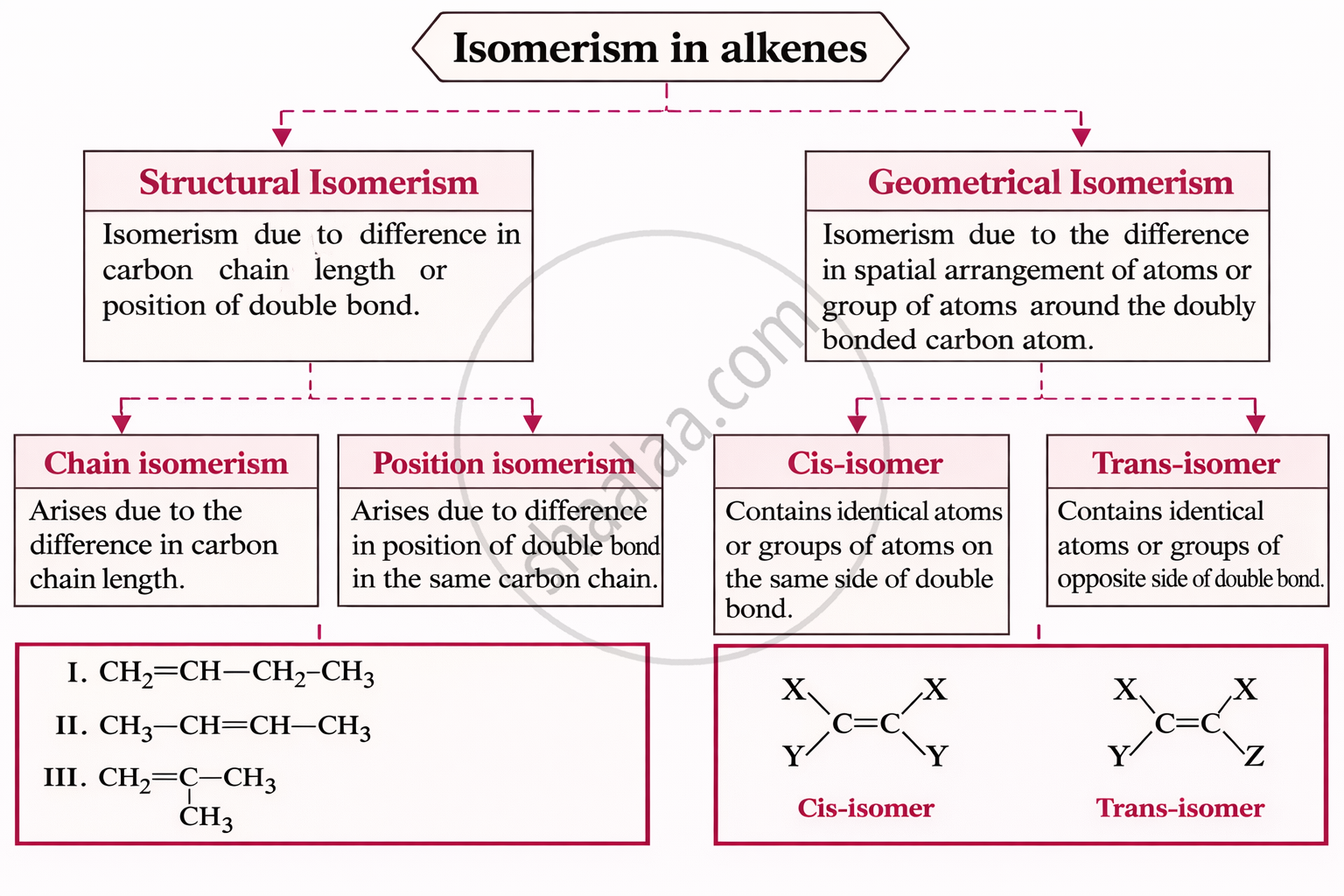
| Method | Example |
|---|---|
| Dehydration of alcohol | \[\ce{CH3CH2CH2OH ->[Conc H2SO4][160°C] CH3CH = CH2 + H2O}\] |
| Dehydrohalogenation of alkyl halide | \[\begin{array}{cc} \phantom{}\ce{CH3 - CH2 ->[Alc KOH][Δ] CH2 = CH2 + KBr + H2}\phantom{}\\ |\phantom{.............................................}\\ \ce{Br}\phantom{...........................................} \end{array}\] |
| Dehalogenation of vicinal dihalide | 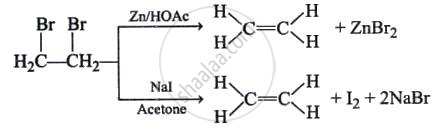 |
| From alkyne (partial reduction — gives cis) |
\[\ce{CH ≡ CH + H2 ->[Pd/C] \underset{cis-alkene}{CH2 = CH2}}\] |
| From alkyne (gives trans) | \[\begin{array}{cc} \phantom{..........................................}\ce{R}\phantom{...........}\ce{H}\phantom{.}\\ \phantom{..........................................}\backslash\phantom{........}/\phantom{.}\\ \phantom{}\ce{R - C ≡ C - R ->[Na][Liquid NH3] C = C}\phantom{}\\ \phantom{..........................................}/\phantom{........}\backslash\phantom{.}\\ \phantom{..........................................}\underset{\text{trans-alkene}}{\ce{R}\phantom{...........}\ce{R}}\phantom{.} \end{array}\] |
1. Solubility
- Alkenes are non-polar
- Insoluble in water
- Soluble in non-polar organic solvents (e.g., hexane, benzene)
2. Boiling Point (BP)
Increases with molecular mass
- More electrons → stronger London dispersion forces
Straight-chain > Branched-chain
- Straight chains have larger surface area → stronger intermolecular forces
Cis-alkenes > Trans-alkenes (usually)
- Cis is more polar → dipoles do not cancel → higher BP
Alkenes vs Alkanes (same number of carbons)
- Alkenes have slightly lower BP
Reason:
- π-bond leads to less effective electron distribution for dispersion forces
- Slightly weaker intermolecular attractions
3. Melting Point (MP)
Trans-alkenes > Cis-alkenes
- Trans is more symmetrical → packs better in crystal lattice → higher MP
Cis-alkenes
- Less symmetrical → poorer packing → lower MP
In trans-alkenes:
- Bond dipoles cancel → non-polar
- Leads to tighter packing in solid state
Alkenes undergo mainly electrophilic addition reactions due to the π bond (electron-rich site).
| Reaction | Example |
|---|---|
| Addition of hydrogen (Hydrogenation) |
\[\ce{H2C = CH2 ->[H2/Ni, Pt or Pd][523-573K] H3C - CH3}\] |
| Addition of halogen | \[\begin{array}{cc} \phantom{}\ce{H3CCH = CH2 + Cl — Cl ->[CCl4] CH3CH - CH2}\phantom{}\\ \phantom{....................................................}|\phantom{.........}|\phantom{}\\ \phantom{.....................................................}\ce{Cl}\phantom{.......}\ce{Cl}\phantom{} \end{array}\] |
| Addition of HX (Markovnikov's rule) | \[\begin{array}{cc} \phantom{..............................................................}\ce{Br}\phantom{}\\ \phantom{............................................................}|\phantom{}\\ \phantom{}\ce{\underset{(For unsymmetrical allkene-Markownikoff’s rule)}{H3CCH = CH2 + HBr} -> H3C - CH - CH3}\phantom{} \end{array}\] |
| Addition of HBr (Anti-Markovnikov / Kharasch effect) |
\[\begin{array}{cc} |
| Hydration (addition of H₂SO₄/H₂O) | \[\begin{array}{cc} \ce{O}\phantom{..}\\ ||\phantom{..}\\ \phantom{}\ce{CH2 = CH2 + H - O - S - O - H -> C2H5HSO4}\phantom{}\\ ||\phantom{..}\\ \ce{O}\phantom{..} \end{array}\] |
| Oxidation (KMnO₄/H⁺) | \[\begin{array}{cc} \phantom{..........................}\ce{O}\phantom{}\\ \phantom{..........................}||\\ \phantom{}\ce{H3C — CH = CH2 ->[{[O]}][KMnO4, {[H^{+}]}] H3C - C - OH + CO2 + H2O}\phantom{} \end{array}\] |
| Hydroxylation | \[\begin{array}{cc} \phantom{}\ce{H2C = CH2 + H2O + [O] ->[Dil.KMnO4][273K] CH2 - CH2}\\ \phantom{.....................................................}|\phantom{..........}|\phantom{}\\ \phantom{........................................................}\ce{OH}\phantom{.....}\ce{OH}\phantom{} \end{array}\] |
| Ozonolysis | \[\begin{array}{cc} \phantom{.....}\ce{H3C}\phantom{....................................}\ce{H3C}\phantom{............................}\\ \phantom{.....}\backslash\phantom{.........................................}\backslash\phantom{.....................}\\ \phantom{..........}\ce{C = CH2 + O3 ->[Zn/H2O] \phantom{.......}C = O + HCHO}\phantom{}\\ \phantom{......}/\phantom{..........................................}/\phantom{.....................}\\ \phantom{...............}\ce{H3C}\phantom{......................................}\ce{H3C}\phantom{......................................} \end{array}\] |
| Polymerisation | |
| Hydroboration-oxidation | \[\begin{array}{cc} \phantom{...............}\ce{H}\phantom{....}\ce{H}\phantom{.............................................}\ce{H}\phantom{....}\ce{H}\phantom{..........................}\ce{H}\phantom{....}\ce{H}\phantom{............................}\\ \phantom{.............}|\phantom{......}|\phantom{..............................................}|\phantom{......}|\phantom{............................}|\phantom{......}|\phantom{..........................}\\ \phantom{}\ce{6(H - C = C - H) + (BH3)2 ->[THF] 2(H - C - C)3 - B ->[H2O2][OH^Θ] H - C - C - H + B(OH)3}\phantom{}\\ \phantom{..........................................}|\phantom{......}|\phantom{............................}|\phantom{......}|\\ \phantom{..............................................}\ce{H}\phantom{.....}\ce{H}\phantom{..........................}\ce{H}\phantom{.....}\ce{OH}\phantom{.} \end{array}\] |
- Used in synthesis of alcohols, plastics, detergents, fuels
- Used for artificial ripening of fruits (like mangoes) — ethylene gas
- General formula: CₙH₂ₙ₋₂
- Suffix for IUPAC naming: –yne
- e.g., Propyne (CH₃–C≡CH), Butyne (CH₃–CH₂–C≡CH)
- C₂H₂ is acetylene (common name); IUPAC name is ethyne
- The triple bond consists of one σ bond and two π bonds
Isomerism in Alkynes:
- Alkynes show position isomerism (type of structural isomerism)
- e.g., 1-Butyne and 2-Butyne
From calcium carbide:
By the action of water on calcium carbide, Cu2C2 or BaC2, acetylene is formed.
CaC2 + 2H2O → CH ≡ CH + Ca(OH)2
\[\ce{CaCO3 ->[][\underset{}{-CO2}] CaO->[2C + Heat][\underset{}{}] CaC2->[H2O][\underset{}{}] CH ≡ CH}\]
Dehalogenation of 1,1,2,2-tetrahaloalkanes:
On treatment with Zn, tetrahalides get dehalogenated to give alkynes.
\[\ce{R - CBr2 - CHBr2 + 2Zn ->[Δ] R - \underset{Alkyne}{C ≡ CH} + 2ZnBr2}\]
\[\ce{R - CBr2 - CBr2 - R + 2Zn ->[Δ] R - \underset{Alkyne}{C ≡ C} - R + 2ZnBr2}\]
By dehydrohalogenation of vic and gem dihalides:
Alkynes are prepared by dehydrohalogenation of vic. and gem. dihalides with alc. KOH + NaNH2 or KNH2.
\[\ce{R - \underset{β}{C}H2 - CHX2 ->[(i) NaNH2/Δ][(ii) H^{+}] R - C ≡ CH}\]
\[\begin{array}{cc}
\ce{X}\phantom{.......................}\\
\phantom{}|\phantom{.......................}\\
\phantom{}\ce{R - \underset{β}{C}H2 - C - R ->[NaNH2/Δ] R - C ≡ CH}\phantom{}\\
\phantom{}|\phantom{.......................}\\
\ce{X}\phantom{.......................}
\end{array}\]
Odour: Alkynes are generally odourless, but acetylene smells of garlic due to phosphine impurity
Boiling and melting points: Slightly higher than those of corresponding alkenes and alkanes with similar carbon atoms
| Order | BP/MP Comparison |
|---|---|
| BP of alkynes | Hex-1-yne > Pent-1-yne > But-1-yne > Propyne > Ethyne |
| MP of alkynes | Ethyne > Pent-1-yne > Propyne > But-1-yne > Hex-1-yne |
Reason for higher BP: Linear structure around the triple bond allows electrons to come closer together, resulting in greater London forces
Solubility: Alkynes are soluble in organic solvents like benzene, CCl₄, and ether
Density: Increases with increase in molecular size
Non-polar molecules → insoluble in water
| Reaction | Examples |
|---|---|
| Acidity of terminal alkynes |
\[\ce{HC ≡ CH + Na -> HC ≡ C^⊖Na^{+} + 2H2}\] |
| Addition of hydrogen (Hydrogenation) |
\[\ce{CH3C ≡ CH + 2H2 ->[Ni] CH3CH2CH3}\] |
| Addition of halogen | \[\begin{array}{cc} \phantom{..........................}\ce{Br}\phantom{...}\ce{Br}\phantom{}\\ \phantom{.........................}|\phantom{......}|\phantom{}\\ \phantom{}\ce{HC ≡ CH ->[2Br2][CCl4] H - C - C - H}\phantom{}\\ \phantom{.........................}|\phantom{......}|\phantom{}\\ \phantom{..........................}\ce{Br}\phantom{...}\ce{Br}\phantom{} \end{array}\] |
| Addition of HX |
\[\ce{HC ≡ CH + 2HBr -> CH3CHBr2}\] |
| Hydration (Hydration of alkynes) | \[\ce{HC ≡ CH + H2O ->[1\% HgSO4][40\% H2SO4] [\underset{Unstable}{CH2 = CHOH}] ⇌ \underset{\underset{compound}{Carbonyl}}{CH3CHO}}\] |
- Used in the manufacture of polymers, synthetic rubber, and plastics
- Used in the artificial ripening of fruits
- Used in welding and cutting of metals (oxy-acetylene flame, temperature ~3500°C)
Cyclic, planar hydrocarbons with delocalised π electrons.
Benzenoids contain benzene ring; Non-benzenoids are aromatic without benzene ring.
Structure of Benzene:
- All 6 C atoms are sp² hybridised
- Unhybridised p-orbitals overlap laterally → delocalised π bonds
- Bond length = 139 pm (uniform, due to resonance)
Huckel's Rule
- Cyclic and planar
- Each ring atom has a p-orbital
- Contains (4n + 2) π electrons (n = 0, 1, 2…)
Examples: Benzene, Naphthalene, Pyridine.
Physical Properties:
- Colourless liquid, sweet smell
- BP = 353 K, MP = 278.5 K
- Immiscible with water; burns with sooty flame
Preparation:
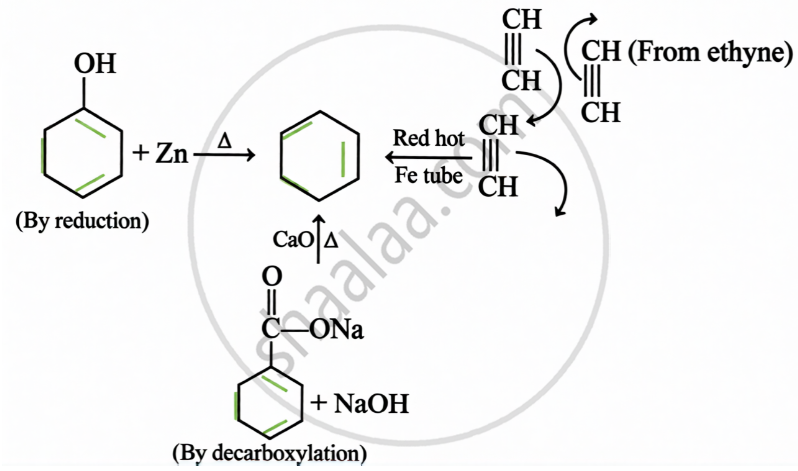
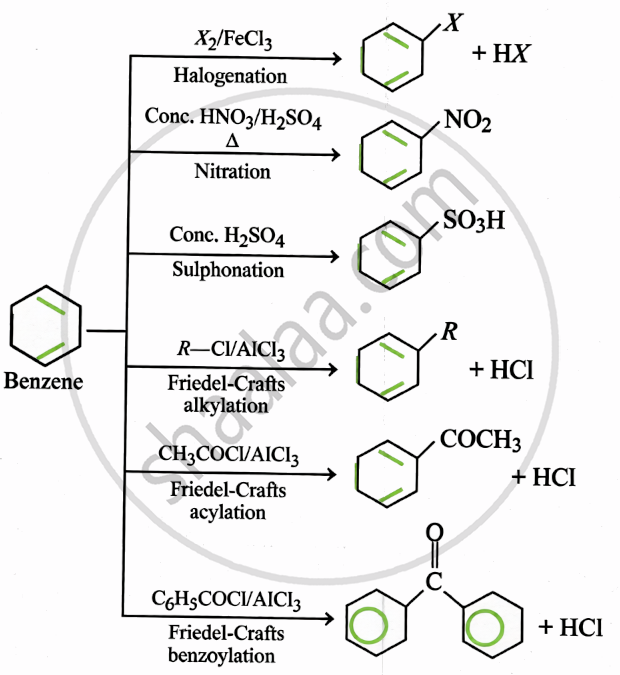
Electrophilic Substitution:
Concepts [16]
- Alkanes
- Preparation of Alkanes
- Physical Properties of Alkanes
- Chemical Properties of Alkanes
- Uses of Alkanes
- Alkenes
- Preparation of Alkenes
- Physical Properties of Alkenes
- Chemical Properties of Alkenes
- Uses of Alkenes
- Alkynes
- Preparation of Alkynes
- Physical Properties of Alkynes
- Chemical Properties of Alkynes
- Uses of Alkynes
- Aromatic Hydrocarbons
