Definitions [1]
Definition: Alkenes
Alkenes are unsaturated hydrocarbons containing at least one C=C double bond.
Key Points
Key Points: Alkenes
- General formula: CₙH₂ₙ (where n = 2, 3, 4…)
- Suffix for IUPAC naming: –ene
- e.g., Ethene (CH₂=CH₂), Propene (CH₃–CH=CH₂)
- The double bond consists of one σ bond and one π bond
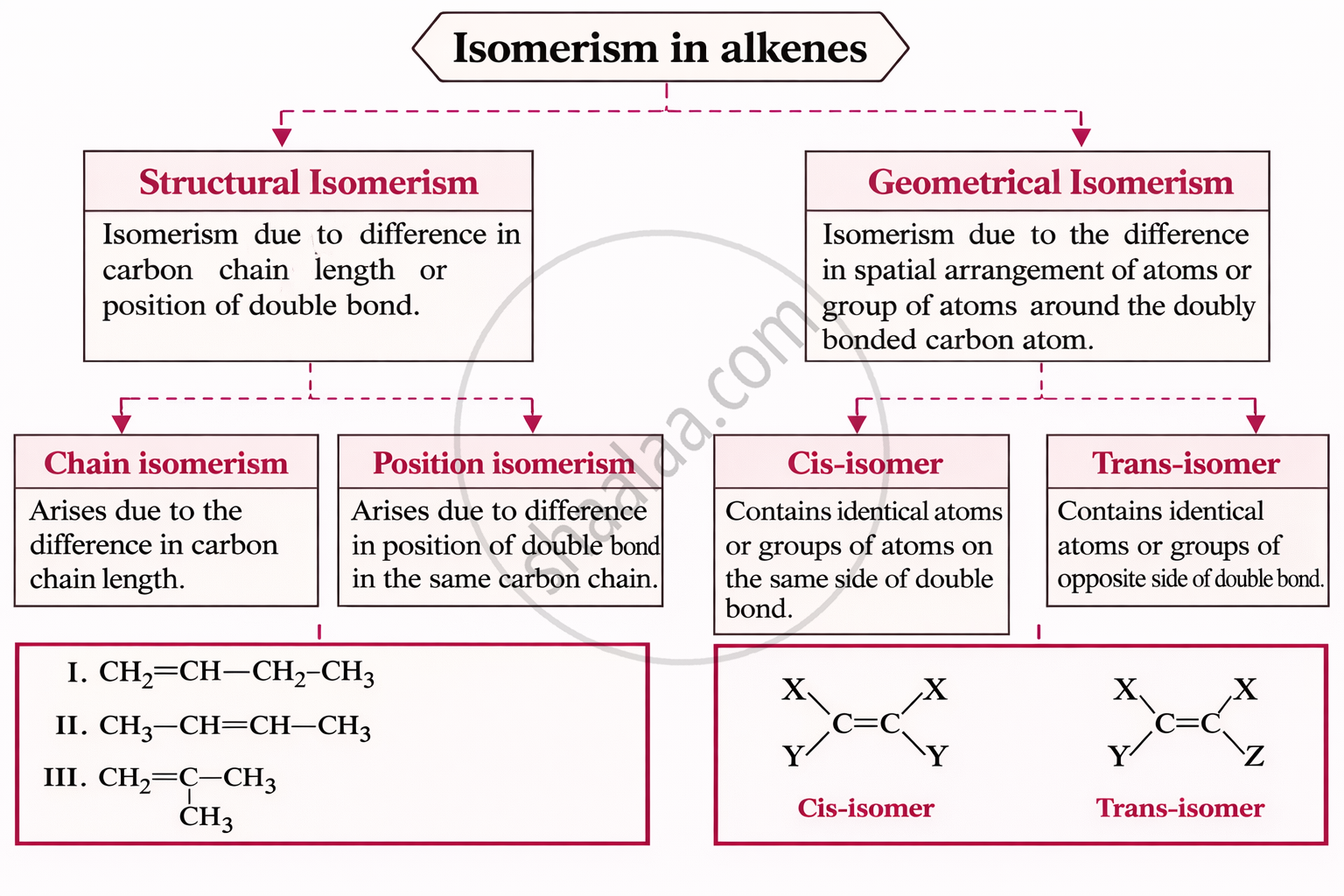
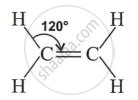
Key Points: Ethene
- Ethene (C₂H₄) is the first member of the alkene series, found in natural gas and acts as a plant hormone responsible for fruit ripening.
- Each carbon in ethene forms two C–H single bonds and one C=C double bond, making a planar (flat) molecule.
- The bond angles in ethene (H–C–H and H–C=C) are approximately 120°, showing trigonal planar geometry.
- Structure of Ethene:
Key Points: Physical Properties of Alkenes
1. Solubility
- Alkenes are non-polar
- Insoluble in water
- Soluble in non-polar organic solvents (e.g., hexane, benzene)
2. Boiling Point (BP)
Increases with molecular mass
- More electrons → stronger London dispersion forces
Straight-chain > Branched-chain
- Straight chains have larger surface area → stronger intermolecular forces
Cis-alkenes > Trans-alkenes (usually)
- Cis is more polar → dipoles do not cancel → higher BP
Alkenes vs Alkanes (same number of carbons)
- Alkenes have slightly lower BP
Reason:
- π-bond leads to less effective electron distribution for dispersion forces
- Slightly weaker intermolecular attractions
3. Melting Point (MP)
Trans-alkenes > Cis-alkenes
- Trans is more symmetrical → packs better in crystal lattice → higher MP
Cis-alkenes
- Less symmetrical → poorer packing → lower MP
In trans-alkenes:
- Bond dipoles cancel → non-polar
- Leads to tighter packing in solid state
Key Points: Chemical Properties of Alkenes
Alkenes undergo mainly electrophilic addition reactions due to the π bond (electron-rich site).
| Reaction | Example |
|---|---|
| Addition of hydrogen (Hydrogenation) |
\[\ce{H2C = CH2 ->[H2/Ni, Pt or Pd][523-573K] H3C - CH3}\] |
| Addition of halogen | \[\begin{array}{cc} \phantom{}\ce{H3CCH = CH2 + Cl — Cl ->[CCl4] CH3CH - CH2}\phantom{}\\ \phantom{....................................................}|\phantom{.........}|\phantom{}\\ \phantom{.....................................................}\ce{Cl}\phantom{.......}\ce{Cl}\phantom{} \end{array}\] |
| Addition of HX (Markovnikov's rule) | \[\begin{array}{cc} \phantom{..............................................................}\ce{Br}\phantom{}\\ \phantom{............................................................}|\phantom{}\\ \phantom{}\ce{\underset{(For unsymmetrical allkene-Markownikoff’s rule)}{H3CCH = CH2 + HBr} -> H3C - CH - CH3}\phantom{} \end{array}\] |
| Addition of HBr (Anti-Markovnikov / Kharasch effect) |
\[\begin{array}{cc} |
| Hydration (addition of H₂SO₄/H₂O) | \[\begin{array}{cc} \ce{O}\phantom{..}\\ ||\phantom{..}\\ \phantom{}\ce{CH2 = CH2 + H - O - S - O - H -> C2H5HSO4}\phantom{}\\ ||\phantom{..}\\ \ce{O}\phantom{..} \end{array}\] |
| Oxidation (KMnO₄/H⁺) | \[\begin{array}{cc} \phantom{..........................}\ce{O}\phantom{}\\ \phantom{..........................}||\\ \phantom{}\ce{H3C — CH = CH2 ->[{[O]}][KMnO4, {[H^{+}]}] H3C - C - OH + CO2 + H2O}\phantom{} \end{array}\] |
| Hydroxylation | \[\begin{array}{cc} \phantom{}\ce{H2C = CH2 + H2O + [O] ->[Dil.KMnO4][273K] CH2 - CH2}\\ \phantom{.....................................................}|\phantom{..........}|\phantom{}\\ \phantom{........................................................}\ce{OH}\phantom{.....}\ce{OH}\phantom{} \end{array}\] |
| Ozonolysis | \[\begin{array}{cc} \phantom{.....}\ce{H3C}\phantom{....................................}\ce{H3C}\phantom{............................}\\ \phantom{.....}\backslash\phantom{.........................................}\backslash\phantom{.....................}\\ \phantom{..........}\ce{C = CH2 + O3 ->[Zn/H2O] \phantom{.......}C = O + HCHO}\phantom{}\\ \phantom{......}/\phantom{..........................................}/\phantom{.....................}\\ \phantom{...............}\ce{H3C}\phantom{......................................}\ce{H3C}\phantom{......................................} \end{array}\] |
| Polymerisation | |
| Hydroboration-oxidation | \[\begin{array}{cc} \phantom{...............}\ce{H}\phantom{....}\ce{H}\phantom{.............................................}\ce{H}\phantom{....}\ce{H}\phantom{..........................}\ce{H}\phantom{....}\ce{H}\phantom{............................}\\ \phantom{.............}|\phantom{......}|\phantom{..............................................}|\phantom{......}|\phantom{............................}|\phantom{......}|\phantom{..........................}\\ \phantom{}\ce{6(H - C = C - H) + (BH3)2 ->[THF] 2(H - C - C)3 - B ->[H2O2][OH^Θ] H - C - C - H + B(OH)3}\phantom{}\\ \phantom{..........................................}|\phantom{......}|\phantom{............................}|\phantom{......}|\\ \phantom{..............................................}\ce{H}\phantom{.....}\ce{H}\phantom{..........................}\ce{H}\phantom{.....}\ce{OH}\phantom{.} \end{array}\] |
Key Points: Uses of Ethene
- Used to make polythene and synthetic chemicals.
- Converted to ethanol for cosmetics and toiletries.
- Helps in fruit ripening.
- Used in epoxyethane and oxy-ethylene flame for detergents and metal cutting.
