Definitions [37]
The equation that combines Boyle's Law, Charles' Law, and Gay-Lussac's Law into a single relation for a fixed mass of gas, relating the quantities pressure (P), volume (V), and temperature (T) which describe the state of the gas, is called the Equation of State.
A gas whose molecules are identical, spherical, rigid, and perfectly elastic point masses, which keep colliding among themselves and with the walls of the containing vessel in perfectly elastic collisions (total energy before collision = total energy after collision), and between which no attractive or repulsive force acts, is called an ideal gas.
Define mean free path and write down its expression.
The average distance travelled by the molecule between collisions is called mean free path (λ).
λ = `"kT"/(sqrt(2)π"d"^2"p")`
The average distance travelled by a gas molecule between two successive collisions is called the mean free path.
OR
The distance travelled by a gas molecule between two successive collisions, during which it moves in a straight line with constant velocity, is called free path.
The square root of the mean of the squares of the speeds of different molecules of a gas is called Root Mean Square Speed (vrms).
The temperature at which the rms speed of molecules of a gas becomes zero (i.e., T = 0 K, vrms = 0) is called absolute zero.
The energy possessed purely by the motion of molecules in an ideal gas, where the molecules are non-interacting and hence there is no potential energy term, making the internal energy of the gas entirely kinetic in nature, is called the kinetic energy (internal energy) of an ideal gas.
The certain minimum value of temperature below which an object cannot be cooled, since the average kinetic energy of molecules has a minimum possible value of zero at this point, is called absolute zero.
The square root of the mean of squares of the speeds of all the molecules of a gas at a given temperature is called root mean square speed.
The law which states that for any system in thermal equilibrium, the total energy is equally distributed among all its degrees of freedom, with energy \[\frac {1}{2}\]kT associated with each degree of freedom per molecule, is called the Law of Equipartition of Energy.
Define heat capacity.
The heat capacity of a body is the quantity of heat required to raise its temperature by 1°C. It depends upon the mass and the nature of the body.
The amount of heat required to raise the temperature of one mole of a substance through a unit degree Celsius or Kelvin is called molar heat capacity.
The quantity of heat needed to raise the temperature of the whole body by 1°C (or 1 K) is called heat capacity.
OR
The amount of heat ΔQΔQ supplied to a substance to change its temperature from T to T + ΔT, per unit mass per unit degree change in temperature, is called specific heat:
-
Unit: J kg⁻¹ K⁻¹
The specific heat capacity of a substance is the amount of heat energy required to raise the temperature of unit mass of that substance through 1°C (or 1 K).
OR
Heat capacity of a body when expressed for the unit mass is called the specific heat capacity of the substance of that body.
OR
The amount of heat energy required to raise the temperature of a unit mass of an object by 1 °C is called the specific heat of that object.
OR
The amount of heat per unit mass absorbed or given out by a substance to change its temperature by one unit (one degree), i.e., 1°C or 1 K, is called specific heat capacity.
OR
The quantity of heat required to raise the temperature of a unit mass of a gas by one degree, whose exact value depends upon the mode of heating the gas and can range from zero to infinity or even be negative, is called the specific heat capacity of a gas.
The ratio of the amount of thermal radiations reflected (QrQr) by a body in a given time to the total amount of thermal radiations incident on the body in that time is called reflectance or reflecting power.
Define diathermanous substance.
Diathermanous substances that allow transmission of infrared radiation through them are called diathermanous substances.
For example - rock salt, pure air, glass, etc.
Define athermanous substance.
Athermanous substances that don't allow transmission of infrared radiation through them are called athermanous substances.
For example - wood, metal, CO2, water, benzene, etc.
The ratio of the amount of thermal radiations absorbed (QaQa) by a body in a given time to the total amount of thermal radiations incident on the body in that time is called absorptance or absorbing power.
The ratio of the amount of thermal radiations transmitted (QtQt) by a body in a given time to the total amount of thermal radiations incident on the body in that time is called transmittance or transmitting power.
A body which absorbs the entire radiant energy incident on it, neither reflecting nor transmitting any radiation, so that its absorptance is unity (a = 1, t = 0, r = 0), and which appears black in light and glows in the dark because a good absorber is always a good emitter, is called a perfectly black body.
A body that emits heat radiations at all finite temperatures (except 0 K) while simultaneously absorbing radiations from its surroundings via radiation is called emission of heat radiation.
Define Boltzmann constant.
It is the molar gas constant (R) to Avogadro constant (Avogadro number) ratio NA.
A real gas is a gas whose molecules interact with each other and therefore does not obey the ideal gas equation under all conditions.
A real gas behaves like an ideal gas at low pressure and high temperature when intermolecular forces become negligible.
The mean free path is the average distance travelled by a gas molecule between two successive collisions.
Degrees of freedom of a system are defined as the total number of coordinates or independent quantities required to describe the position and configuration of the system completely.
Radiation emitted by a heated cavity is called cavity radiation and depends only on the temperature of the cavity walls.
Thermal radiation is the electromagnetic radiation emitted by a body due to its temperature.
The ratio of amount of heat absorbed to total quantity of heat incident is called the coefficient of absorption.
The ratio of the amount of radiant energy reflected to the total energy incident is called the coefficient of reflection.
The ratio of amount of radiant energy transmitted to total energy incident is called the coefficient of transmission.
A body, which absorbs the entire radiant energy incident on it, is called an ideal or perfect blackbody.
A blackbody is a body that absorbs all incident radiation of all wavelengths and does not reflect or transmit any radiation.
(Absorptivity a = 1, reflectivity r = 0, transmissivity t = 0)
Ferry’s blackbody is a practical model of a perfect blackbody consisting of a hollow cavity with a small aperture that absorbs almost all incident radiation.
Radiation is the mode of transfer of heat in the form of electromagnetic waves without requiring a material medium.
A gas that obeys the equation PV = nRT at all pressures and temperatures is called an ideal gas.
The equation relating pressure, volume, temperature and other state variables of a gas is called the equation of state.
Formulae [15]
Combining the above three laws for a fixed mass of gas:
PV = nRT
PV = NkBT
where:
- P = pressure, V = volume, T = temperature
- n = number of moles, R = universal gas constant
- N = number of molecules, kB = Boltzmann's constant
Let λ1, λ2, λ3,…λn be the distances travelled by a gas molecule during nn collisions respectively, then the mean free path is:
-
Inversely with the density (ρ) of the gas:
λ ∝ \[\frac {1}{ρ}\] -
Inversely with the square of the diameter (dd) of the molecule:
λ ∝ \[\frac {1}{d^2}\]
\[v_{rms}=\sqrt{\frac{v_1^2+v_2^2+v_3^2+v_4^2+\ldots}{N}}\]
where v1, v2, v3… are speeds of individual molecules and N = total number of molecules.
\[Q=mc\Delta T\]
Specific heat capacity c = \[\frac{\text{Heat capacity of body } C'}{\text{Mass of the body } m}\]
or
Specific heat capacity c = \[\frac{Q}{m\times\Delta t}\]
C = M × c = Q/(nΔT)
Unit: J/mol · K
a = \[\frac {Q_a}{Q}\] r = \[\frac {Q_r}{Q}\] tr = \[\frac {Q_t}{Q}\]
where Q = total heat/energy incident on the surface of the object.
Key Relation: a + r + tr = 1
P ∝ T (at constant V)
V ∝ T (at constant P)
V ∝ \[\frac {1}{P}\] (at constant T)
\[\lambda=\frac{1}{\sqrt{2}\pi d^2\left(\frac{N}{V}\right)}\]
Where:
- λ = mean free path
- d = diameter of molecule
- N/V = number density of molecules
P =\[\frac{1}{3}\frac{N}{V}m\overline{\mathrm{v}^2}\]
Vs = \[\sqrt{\frac{\gamma RT}{M_{0}}}\]
where,
γ = \[\frac {C_p}{C_v}\]
\[\frac{1}{2}m\overline{v^2}=\frac{3}{2}k_BT\]
Cp - Cv = R
Theorems and Laws [15]
At constant temperature, the volume of a fixed mass of gas is inversely proportional to its pressure.
V ∝ \[\frac {1}{P}\]
At constant pressure, the volume of a fixed mass of gas is directly proportional to its temperature.
V ∝ T(at constant P)
At constant volume, the pressure of a fixed mass of gas is directly proportional to its temperature.
P ∝ T(at constant V)
What is an ideal gas?
An ideal or perfect gas is a gas which obeys the gas laws (Boyle’s law, Charles’ law, and Gay-Lussac’s law) at all pressures and temperatures. An ideal gas cannot be liquefied by the application of pressure or by lowering the temperature.
RMS speed decreases with increase in molecular weight:
RMS speed varies directly with the square root of temperature:
With rise in temperature, vrms of gas molecules increases.
The average energy per molecule of an ideal gas is directly proportional to the absolute temperature T of the gas:
Statement:
For a gas in thermal equilibrium at temperature TT, the average energy associated with each molecule for each quadratic term (degree of freedom) is:
where kB = 1.38 × 10−23 J/K and T = absolute temperature.
Energy Expressions for Different Types of Motion:
-
Translational K.E.:
\[\frac{1}{2}mv_x^2+\frac{1}{2}mv_y^2+\frac{1}{2}mv_z^2\] (3 degrees of freedom — along x, y, z axes) -
Rotational K.E.:
\[\frac{1}{2}I\omega_x^2+\frac{1}{2}I\omega_y^2+\frac{1}{2}I\omega_z^2\] (up to 3 degrees of freedom — rotation about x, y, z axes) -
Vibrational K.E.:
\[\frac{1}{2}m\dot{u}^2+\frac{1}{2}kr^2\] (2 terms — kinetic and potential energy of vibration)
Each quadratic term contributes \[\frac {1}{2}\]kBT to the total average energy of the molecule.
State and prove Kirchoff’s law of heat radiation.
Kirchhoff’s law of thermal radiation deals with wavelength specific radiative emission and absorption by a body in thermal equilibrium. It states that at a given temperature, the ratio of emissive power to coefficient of absorption of a body is equal to the emissive power of a perfect blackbody at the same temperature for all wavelengths.
Since we can describe the emissive power of an ordinary body in comparison to a perfect blackbody through its emissivity, Kirchhoff’s law can also be stated as follows: for a body emitting and absorbing thermal radiation in thermal equilibrium, the emissivity is equal to its absorptivity.
Symbolically, a = e or more specifically a(λ) = e(λ).
Thus, if a body has high emissive power, it also has high absorptive power and if a body has low emissive power, it also has low absorptive power.
Kirchhoff’s law can be theoretically proved by the following thought experiment. Consider an ordinary body A and a perfect blackbody B of identical geometric shapes placed in an enclosure. In thermal equilibrium, both bodies will be at same temperature as that of the enclosure.
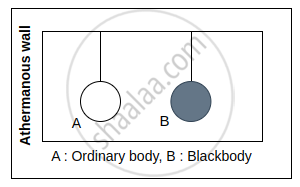
Let R be the emissive power of body A, RB be the emissive power of blackbody B and a be the coefficient of absorption of body A. If Q is the quantity of radiant heat incident on each body in unit time and Qa is the quantity of radiant heat absorbed by the body A, then Qa = a Q. As the temperatures of the body A and blackbody B remain the same, both must emit the same amount as they absorb in unit time. Since emissive power is the quantity of heat radiated from unit area in unit time, we can write
Quantity of radiant heat absorbed by body A = Quantity of heat emitted by body A
or a Q = R ...(1)
For the perfect blackbody B,
Q = RB ...(2)
From Eqs. (1) and (2), we get,
a = `R/Q = R/R_b` ...(3)
From Eq. (3), we get, `R/a = R_b`
By definition of coefficient of emission,
`R/R_b` ...(4)
From Eqs. (3) and (4), we get, a = e.
Hence, the proof of Kirchhoff‘s law of radiation.
At a given temperature, the ratio of emissive power to the coefficient of absorption of a body is equal to the emissive power of a perfect blackbody at the same temperature for all wavelengths:
The wavelength (λm) for which the emissive power of a blackbody is maximum is inversely proportional to the absolute temperature of the blackbody:
With increase in temperature, λm decreases (shifts towards shorter wavelengths). Also, the energy Emax emitted at λm increases with the fifth power of temperature, i.e., Emax ∝ T5.
The rate of emission of radiant energy per unit area (power radiated per unit area) of a perfect blackbody is directly proportional to the fourth power of its absolute temperature:
The area under the spectral curve increases with temperature and is directly proportional to T4.
It states that at a given temperature, the ratio of emissive power to coefficient of absorption of a body is equal to the emissive power of a perfect blackbody at the same temperature for all wavelengths.
It is observed that the wavelength, for which the emissive power of a blackbody is maximum, is inversely proportional to the absolute temperature of the blackbody. This is Wien's displacement law.
λmax T = b
b = Wien's constant
According to this law, "The rate of emission of radiant energy per unit area or the power radiated per unit area of a perfect blackbody is directly proportional to the fourth power of its absolute temperature".
R = eσT4
Key Points
- In gases, the intermolecular forces are very weak, causing the molecules to move apart in all directions.
- Gases have no fixed shape and no fixed size — they can be obtained in a vessel of any shape or size.
- Gases expand indefinitely and uniformly to fill any available space.
- Gases exert pressure on their surroundings.
- Independence from Pressure: vrms does not depend on the pressure of the gas at constant temperature. If pressure is increased n times, density also increases n times, but vrms remains constant (Boyle's Law: p ∝ ρ).
- Atmosphere of Planets: A planet or satellite will have an atmosphere only if vrms < ve (escape velocity). Moon has no atmosphere because vrms of its gas molecules is greater than escape velocity (ve).
- Heat energy absorbed (Q) depends on: mass (m), rise in temperature (Δt), and specific heat capacity (c), i.e., Q ∝ m × Δt × c.
- Heat capacity (C') and specific heat capacity (c) are related by: C′ = m × c.
- A perfectly black body absorbs completely the radiations of all wavelengths incident on it — it neither reflects nor transmits any radiation, hence a = 1.
- The colour of an opaque body is the colour (wavelength) of radiation reflected by it. Since a black body reflects no wavelength, it appears black whatever be the colour of radiations incident on it.
- In practice, Ferry's body is considered a perfect black body — the lampblack used in it absorbs 97% of incident energy.
- A perfect black body appears black in light and glows in the dark, because a good absorber is always a good emitter of thermal radiation.
- Every body emits and absorbs heat radiations at all finite temperatures (except 0 K).
- Energy exchange among bodies takes place via radiation.
- If radiation absorbed > emitted → body's temperature increases and it appears hotter.
- At absolute zero (0 K or −273°C), all heat exchange ceases completely.
- A diatomic molecule has 3 translational and 2 rotational degrees of freedom.
- Each degree of freedom contributes \[\frac {1}{2}\]kBTenergy (law of equipartition).
- At high temperatures, vibrational motion also adds extra energy to the molecule.
- Monatomic gas: Has 3 translational degrees of freedom, so
Cv = \[\frac {3}{2}\]R, Cp = \[\frac {5}{2}\]R, and γ = \[\frac {5}{3}\]. - Diatomic gas (rigid): Has 3 translational + 2 rotational degrees of freedom, so
Cv = \[\frac {5}{2}\]R, Cp = \[\frac {7}{2}\]R, and γ = \[\frac {7}{5}\]. - Polyatomic gas: Has translational, rotational, and vibrational degrees of freedom; more degrees of freedom → higher internal energy and specific heat.
- All bodies above 0 K emit and absorb heat radiation continuously.
- If a body emits more than it absorbs, its temperature falls; if it absorbs more, its temperature rises.
- Heat radiated depends on temperature, surface area, surface nature, and time.
- Emissive power is heat radiated per unit area per unit time.
- Emissivity (e) compares a body’s radiation to a blackbody; 0 < e ≤ 1.
- Blackbody radiation has many wavelengths, and its pattern depends only on temperature.
- For a given temperature, intensity rises, reaches a maximum (λₘₐₓ), then decreases.
- As the temperature increases, the peak shifts to shorter wavelengths.
- Higher temperature means more total energy is emitted.
- Classical theory failed to explain this, but Planck’s theory did.
Important Questions [16]
- Choose the correct option. If the pressure of an ideal gas decreases by 10% isothermally, then its volume will ______.
- Choose the correct option. The mean free path λ of molecules is given by where n is the number of molecules per unit volume and d is the diameter of the molecules.
- Derive an expression for the pressure exerted by a gas on the basis of the kinetic theory of gases.
- Compare the rms speed of hydrogen molecules at 227°C with the rms speed of oxygen molecules at 127°C. Given that molecular masses of hydrogen and oxygen are 2 and 32 respectively.
- The root mean square speed of the molecules of a gas is proportional to ______.[T =Absolute temperature of gas]
- Show that the average energy per molecule is directly proportional to the absolute temperature ‘T’ of the gas.
- Compare the rms speed of hydrogen molecules at 127°C with rms speed of oxygen molecules at 27ºC given that molecular masses of hydrogen and oxygen are 2 and 32 respectively.
- In an ideal gas, the molecules possess ______.
- What is the Ratio of Two Specific Heats,Cp/Cv ?
- Prove the Mayer's relation Cp-Cv=RJ
- The difference between the two molar specific heats of gas is 9000 J/kg K. If the ratio of the two specific heats is 1.5, calculate the two molar specific heats.
- Draw a neat labelled diagram of Ferry’s perfectly black body.
- Explain the construction and working of Ferry’s black body.
- State and prove Kirchoff’s law of heat radiation.
- State Wein'S Displacement Law
- Compare the rate of loss of heat from a metal sphere at 827°C with the rate of loss of heat from the same at 427°C, if the temperature of the surrounding is 27°C.
Concepts [16]
- Gases and Its Characteristics
- Classification of Gases: Real Gases and Ideal Gases
- Mean Free Path
- Expression for Pressure Exerted by a Gas
- Root Mean Square (RMS) Speed
- Interpretation of Temperature in Kinetic Theory
- Law of Equipartition of Energy
- Specific Heat Capacity
- Absorption, Reflection, and Transmission of Heat Radiation
- Perfect Blackbody
- Emission of Heat Radiation
- Kirchhoff’s Law of Heat Radiation and Its Theoretical Proof
- Spectral Distribution of Blackbody Radiation
- Wien's Displacement Law
- Stefan-boltzmann Law of Radiation
- Overview: Kinetic Theory of Gases and Radiation
