Definitions [3]
Define cathode
The electrode at which the reduction occur is called cathode.
Define anode
The electrode at which the oxidation occur is called anode.
Define the following term:
Fuel cell
Fuel cells are the galvanic cells in which the energy of combustion of fuels like hydrogen, methanol, etc., is directly converted into electrical energy.
Theorems and Laws [1]
State Faraday’s Laws of electrolysis.
Faraday’s laws of electrolysis:
- First law: The mass of the substance (M) liberated at an electrode during electrolysis is directly proportional to the quantity of charge (Q) passed through the cell, i.e., M ∝ Q.
- Second law: When the same quantity of charge is passed through the solutions of different electrolytes, the amount of substances liberated at the respective electrodes are directly proportional to their electrochemical equivalents, i.e., M ∝ Z.
Faraday’s first law of electrolysis: Faraday’s first law of electrolysis may be stated as follows.
The amount of substance liberated (or deposited) at a particular electrode during electrolysis is directly proportional to the quantity of electricity (total charge) passed in the solution.
W ∝ Q
Since Charge = Current × Time
we have Q = I × t
where I is the current (in amperes) passed in the solution for t seconds. Hence,
W ∝ I × t
or, W = Z × I × t ...(i)
In the above equation, Z is a constant known as the electrochemical equivalent of the substance liberated at the electrode.
If I = 1 ampere and t = 1 s, we have
W = Z
Hence, electrochemical equivalent may be defined as the amount of substance liberated by a current of one ampere passed for one second.
Faraday’s second law of electrolysis: Faraday’s second law of electrolysis can be stated as follows.
When the same quantity of electricity is passed through the solutions of different electrolytes connected in series, the masses of the substances liberated (or deposited) at the electrodes are directly proportional to their equivalent masses.
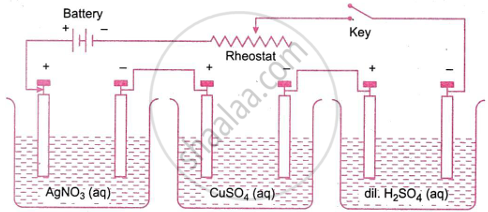
Suppose three voltameters connected in series contain aqueous solutions of silver nitrate, copper sulphate, and dilute sulphuric acid, respectively, as shown in Fig. On passing electric current, electrolysis takes place in all three cells, and the substances produced at the cathodes of these cells are Ag(s), Cu(s), and H2(g), respectively. If the same current is passed for the same time, it is found that
\[\frac{\text{Mass of silver deposited}}{\text{Mass of copper deposited}} = \frac{\text{Equivalent mass of silver}}{\text{Equivalent mass of copper}}\] and,
\[\frac{\text{Mass of hydrogen gas liberated}}{\text{Mass of silver deposited}} = \frac{\text{Equivalent mass of hydrogen}}{\text{Equivalent mass of silver}}\]
Key Points
| Type | Electrolytic Cell | Galvanic (Voltaic) Cell |
|---|---|---|
| Energy conversion | Electrical → Chemical | Chemical → Electrical |
| Nature of reaction | Non-spontaneous | Spontaneous |
| Anode | Positive | Negative |
| Cathode | Negative | Positive |
| Electron flow | Cathode → Anode | Anode → Cathode |
| Salt bridge | Not required | Required |
Electrolysis of NaCl
1. Molten NaCl:
-
Oxidation: Cl⁻ → Cl₂ (gas)
-
Reduction: Na⁺ → Na (metal)
-
Products: Na (cathode), Cl₂ (anode)
2. Aqueous NaCl:
-
Oxidation: Cl⁻ → Cl₂
-
Reduction: H₂O → H₂ + OH⁻
-
Products: H₂ (cathode), Cl₂ (anode), NaOH formed
