Definitions [5]
The phenomenon of emission of electronsfrom metals under the effect of light is called 'photoelectric effect'.
The minimum energy required for the emission of photoelectron from a metal is called the 'work function' of that metal.
The negative potential of P2 (relative to P1) at which the photoelectric current becomes zero is called 'stopping potential' or 'cut-off potential'.
The lowest frequency of light which can emit photoelectrons from a material is called the 'threshold frequency' or 'cut-off frequency' of that material.
The energy crossing per unit area per unit time perpendicular to the direction of propagation of wave is called the intensity of wave.
Formulae [4]
n = \[\frac {Pλ}{h c}\]
m = \[\frac {E}{c^2}\] = \[\frac {hv}{c^2}\] = \[\frac {h}{cλ}\]
p = \[\frac {E}{c}\] = \[\frac {h}{λ}\]
P = nh\[\frac {c}{λ}\]
Theorems and Laws [1]
- The rate of emission of photoelectrons from the surface of a metal varies directly as the intensity of the incident light falling on the surface.
- The maximum kinetic energy of the emitted photoelectrons is independent of the intensity of the incident light.
- The maximum kinetic energy of the photoelectrons increases linearly with an increase in the frequency of the incident light.
- If the frequency of the incident light is below a certain lowest value, then no photoelectron is emitted from the metal. This lowest frequency (threshold frequency) is different for different metals.
- As soon as the light is incident on the surface of the metal, the photoelectrons are emitted instantly; that is, there is no time-lag between the incidence of light and the emission of electrons.
Key Points
- Hertz (1887) observed that ultraviolet light makes electric discharge easier from a metal surface.
- Hallwachs’ experiment showed that current flows only when ultraviolet light strikes the negative plate, not the positive plate.
- J.J. Thomson (1898) proved that light falling on a metal surface causes the emission of electrons.
- Lenard (1900) explained that electrons emitted from the negative plate are attracted to the positive plate, producing current.
- Short-wavelength (high-frequency) light is more effective in producing photoelectric emission than long-wavelength light.
- Photoelectric current increases with incident light intensity when the frequency is kept constant.
- For sufficiently high anode potential, the photoelectric current reaches a maximum (saturation current).
- Stopping potential is independent of light intensity and depends on the maximum kinetic energy of photoelectrons.
- A higher frequency of incident light produces photoelectrons with greater maximum kinetic energy.
- No photoelectric emission occurs below a certain frequency, regardless of the intensity or duration of light.
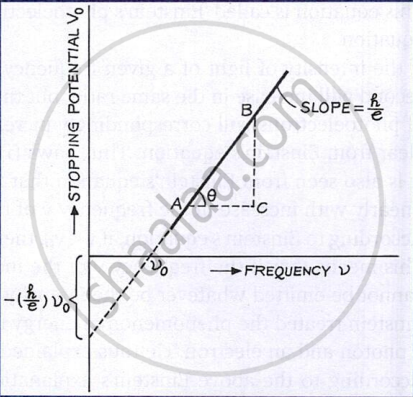
- A graph of stopping potential V0 versus frequency ν is a straight line, showing a linear relation between them.
- The slope of the V0 – ν graph equals h/e, hence Planck’s constant h can be determined using the known value of electronic charge e.
- Radiation behaves like a stream of particles called photons during interaction with matter.
- Photons travel in straight lines at the speed of light.
- Each photon has energy E = hν = \[\frac {hc}{λ}\] and momentum p = \[\frac {E}{c}\].
- On a change of medium, the speed and wavelength of a photon change, but its frequency remains constant.
- Photon energy is independent of light intensity; higher intensity means more photons per second.
- A photon has zero rest mass, but an equivalent mass given by
- m = \[\frac {h}{cλ}\]
- Photons are electrically neutral and are not deflected by electric or magnetic fields.
- In photon–particle collisions, total energy and momentum are conserved.
- A photon retains its identity until absorbed by an atom, after which its identity is lost.
- Wave theory fails because it predicts that electron energy should increase with light intensity, but experiments show that it does not.
- Wave theory cannot explain the existence of a threshold frequency, below which no photoelectrons are emitted.
- Wave theory predicts a time lag in emission, but photoelectrons are emitted instantaneously.
- Wave theory failed to explain experimental observations of the photoelectric effect.
- Black-body radiation contains all wavelengths, and classical theories could not explain its energy distribution.
- Planck proposed that radiation is emitted discontinuously in small energy packets, now known as quanta (photons).
- The energy of a photon is hνh\nuhν, and radiation energy is emitted only in integral multiples of hνh\nuhν.
- Einstein (1905) explained the photoelectric effect using Planck’s photon hypothesis.
- Light consists of photons, each having energy hνh\nuhν; light intensity depends on the number of photons.
- A photon transfers its entire energy to a single electron during photoelectric emission.
- Part of the photon energy is used to overcome the work function, and the rest appears as kinetic energy of the electron.
- Electrons emitted from the metal surface have maximum kinetic energy because they experience no energy loss in collisions.
- Einstein’s photoelectric equation is
Ek = hν − W = h(ν − ν0) - Increasing light intensity increases the number of photoelectrons, but not their maximum kinetic energy.
- Photoelectric emission is instantaneous, and Einstein’s explanation fully accounts for all laws of the photoelectric effect.
