- Carboxylic acids are carbon compounds with a –COOH group and have acidic nature.
- Types:
- Monocarboxylic acids have one –COOH group (e.g., formic acid, acetic acid)
- Dicarboxylic acids have two –COOH groups (e.g., oxalic acid) - IUPAC Naming: Replace the ‘e’ of the corresponding alkane with ‘oic acid’ (e.g., ethane → ethanoic acid); also called alkanoic acids.
Definitions [2]
Definition: Carbonyl Compound
Organic compounds containing carbon-oxygen double bond, i.e. \[\mathrm{>C=O}\] group, are known as carbonyl compounds.
Definition: Carboxylic Acid
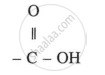
An organic compound containing the carboxyl group (-COOH) is known as carboxylic acid. These compounds possess acidic properties.
Formulae [1]
Formula: Carboxylic Acid
General formula: CnH2n+1COOH (or RCOOH)
Functional group:
Key Points
Key Points: Concepts of Aldehydes, Ketones, and Carboxylic Acids
- Carbonyl group: The ≻C=O group (carbonyl carbon + carbonyl oxygen) — a key functional group in organic chemistry.
- Carbonyl compounds: Aldehydes and ketones, both containing ≻C=O as their functional group.
- Aldehydes: –CHO (formyl group); carbonyl C bonded to at least one H.
- Ketones: ≻C=O (ketonic carbonyl group); carbonyl C bonded to two alkyl/aryl groups (R=R′ or R≠R′).
- Carboxylic acids: –COOH (carboxyl group); –OH attached to ≻C=O makes them distinct from aldehydes/ketones.
Key Points: Chemical Reactions of Aldehydes and Ketones - Nucleophilic Addition Reactions
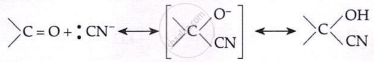
Addition of HCN:
\[\text{HCN} + \text{OH}^- \rightleftharpoons :\text{CN}^- + \text{H}_2\text{O}\]
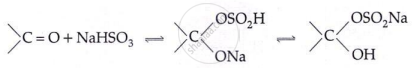
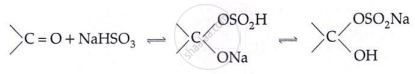
Addition of NaHSO3:
Addition of Grignard reagent:
Addition of alcohols:
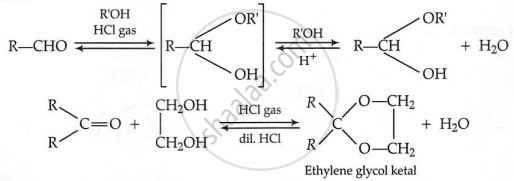
Key Points: Chemical Reactions of Aldehydes and Ketones - Reduction
\[\text{R}-\text{CHO} + 2[\text{H}] \xrightarrow[\text{or BH}_3]{\text{LiAlH}_4} \text{RCH}_2\text{OH}\]
\[\begin{array}{r@{\;}c@{\;}l r@{\;}c@{\;}l} \ce{R} & & & \ce{R} & & \\ & \backslash & & & \backslash & \\ & & \ce{C=O + H2 ->[Ni or Pt]} & & & \ce{CH2OH} \\ & / & & & / & \\ \ce{R} & & & \ce{R} & & \end{array}\]
Clemmensen reduction:
\begin{array}{r@{\;}c@{\;}l c r@{\;}c@{\;}l} & & & & & & \\ & \backslash & & & & \backslash & \\ & & \ce{C=O} & \xrightarrow[\text{HCl}]{\text{Zn-Hg}} & & & \ce{CH2 + H2O} \\ & / & & & & / & \\ & & & & & & \end{array}
Wolff-Kishner reduction:
\begin{array}{r@{\;}c@{\;}l c r@{\;}c@{\;}l c r@{\;}c@{\;}l} & & & & & & & & & & \\ & \backslash & & & & \backslash & & & & \backslash & \\ & & \ce{C=O} & \xrightarrow[-\ce{H2O}]{\ce{NH2NH2}} & & & \ce{C=NNH2} & \xrightarrow{\text{KOH ethylene Glycol, }\Delta} & & & \ce{CH2 + N2} \\ & / & & & & / & & & & / & \\ & & & & & & & & & & \end{array}
Key Points: Chemical Reactions of Aldehydes and Ketones - Oxidation
\[\underset{\text{Aldehyde}}{\ce{R-CHO}} + \ce{[O]} \xrightarrow[\text{or }\ce{KMnO4/H2SO4}]{\ce{K2Cr2O7/H2SO4}} \underset{\text{Carboxylic acid}}{\ce{R-COOH}}\]
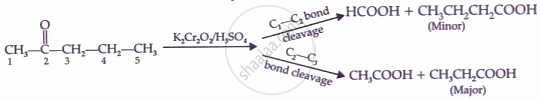
Key Points: Chemical Reactions of Aldehydes and Ketones - Reactions Due to α-hydrogen
Aldol condensation:
\[\ce{CH3 - \overset{\displaystyle O}{\overset{||}{C}} - H + H - CH2 - \overset{\displaystyle O}{\overset{||}{C}} - H} \xrightarrow{\text{dil. NaOH}} \ce{H3C - \underset{\underset{\displaystyle OH}{|}}{CH} - \underset{\underset{\displaystyle H}{|}}{CH} - \overset{\displaystyle O}{\overset{||}{C}} - H} \\ \xrightarrow{\text{dil. }\ce{H2SO4}\Delta, \ce{-H2O}} \ce{CH3 - CH = CH - \overset{\displaystyle O}{\overset{||}{C}} - H}\]
Mechanism:
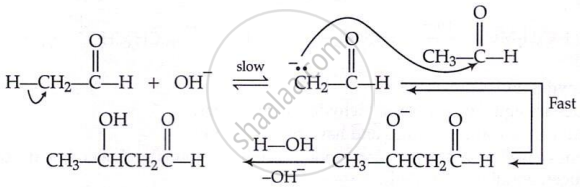
Cross aldol condensation:
\[\begin{array}{ccccccccccccc} & \ce{O} & & \ce{O} & & & & \ce{OH} & & \ce{O} & & & \\ & || & & || & & & & | & & || & & & \\ \ce{C6H5 -} & \ce{C} & \ce{+ HCH2 -} & \ce{C} & \ce{- H} & \overset{\text{dil. NaOH}}{\rightleftharpoons} & \ce{C6H5 -} & \ce{C} & \ce{- CH2 -} & \ce{C} & \ce{- H} & \xrightarrow[\Delta]{\ce{H2O}} & \ce{C6H5CH=CHCHO} \\ & | & & & & & & | & & & & & \text{Cinnamaldehyde} \\ & \ce{H} & & & & & & \ce{H} & & & & & \end{array}\]
Cannizzaro reaction: It is a self-oxidation reduction reaction.
\[\begin{array}{r@{\;}c@{\;}l} \ce{H} & & \\ & \backslash & \\ & & \ce{C=O} \\ & / & \\ \ce{H} & & \end{array} + \begin{array}{r@{\;}c@{\;}l} \ce{H} & & \\ & \backslash & \\ & & \ce{C=O} \\ & / & \\ \ce{H} & & \end{array} + \text{conc. KOH} \xrightarrow{\Delta} \ce{H - \underset{\underset{\displaystyle H}{|}}{\overset{\overset{\displaystyle H}{|}}{C}} - OH} + \ce{H - \overset{\displaystyle O}{\overset{||}{C}} - OK}\]
Haloform Reaction:
The reaction can be used to transform acetyl groups into carboxyl groups or to produce chloroform or iodoform. This reaction has been used in qualitative analysis to indicate the presence of a methyl ketone in which excess iodine is used to halogenate the compound. The product iodoform precipitates as a yellow-coloured substance and has a characteristic odour.
\[\underset{\substack{\text{Acetone} \\ \text{Iodoform Test}}}{\ce{H3C - \overset{\displaystyle O}{\overset{||}{C}} - CH3}} \xrightarrow[4\ce{NaOH}]{3\ce{Cl2}} \underset{\text{Sodium acetate}}{\ce{H3C - \overset{\displaystyle O}{\overset{||}{C}} - O^\ominus Na^\oplus}} + \underset{\text{Chloroform}}{\ce{CHCl3}}\]
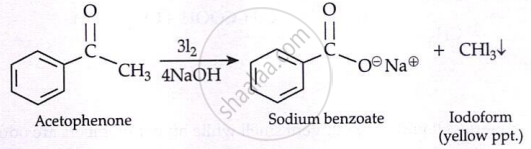
Key Points: Uses of Aldehydes and Ketones
- Formaldehyde: Used in making Bakelite (phenol-formaldehyde resin), as a preservative (formalin = 40% HCHO).
- Acetaldehyde: Used in the preparation of acetic acid and ethanol.
- Acetone: Solvent (nail polish remover), used in the manufacture of chloroform.
- Benzaldehyde: Used in perfumes and dyes.
Key Points: Carboxylic Acid
Key Points: Preparation of Carboxylic Acids
- Special preparations: Benzoic acid from cumene (KMnO₄/KOH, Δ → H₃O⁺); adipic acid from cyclohexene (KMnO₄/dil. H₂SO₄, Δ).
- Aldehyde preparation: From 1° alcohol (K₂Cr₂O₇/H₂SO₄ or Cu/573 K), alkene (ozonolysis), alkyne (dil. H₂SO₄/HgSO₄), acid chloride (Rosenmund), nitrile (Stephen/DIBAL-H); aromatic via Etard, CrO₃/(CH₃CO)₂O, Cl₂/hν, Gatterman–Koch.
- Aldehyde reactions: HCN → cyanohydrin; NaHSO₃ → bisulphite adduct; R′OH → acetal; RMgBr → 2° alcohol; NH₂OH → aldoxime; NH₂NH₂ → hydrazone; K₂Cr₂O₇ → COOH; Clemmensen/Wolf–Kishner → alkane.
- Carboxylic acid preparation: From nitriles (hydrolysis), acyl chloride/anhydride/ester + H₂O, CO₂ + RMgX (dry ether, H₃O⁺), alkylbenzene (KMnO₄–KOH).
- Carboxylic acid reactions: PCl₅/SOCl₂ → RCOCl; NH₃ → amide; P₂O₅ → anhydride; NaOH + CaO (Δ) → alkane; LiAlH₄ → 1° alcohol; ROH/conc. H₂SO₄ → ester.
Key Points: Physical Properties of Carboxylic Acids
- Solubility: Decreases with an increase in the size of the hydrocarbon part.
- Miscibility: Lower carboxylic acids (up to 4 C atoms) are miscible with water due to H-bonding.
- Boiling point: Carboxylic acids have higher B.P. than ketones, aldehydes, and alcohols of comparable molecular mass due to intermolecular H-bonding.
- Order of B.P. (carboxylic acids & aldehydes): Valeric > Butyric > Propionic > Acetic > Formic acid; Hexanal > Pentanal > Butanal > Propanal.
- Order of B.P. (ketones): Hexan-2-one > Pentan-2-one > Butan-2-one > Propanone.
Key Points: Uses of Carboxylic Acids
- Methanoic acid (Formic acid): Leather tanning, dyeing and finishing in textiles.
- Ethanoic acid (Acetic acid): Manufacturing of rayon and plastic; used as vinegar in cooking.
- Benzoic acid: Used as a food preservative and in perfumery.
- Salicylic acid: Used in the preparation of Aspirin (analgesic/antipyretic), Salol, and Oil of Wintergreen (methyl salicylate).
- Aspirin = acetylsalicylic acid; anti-pyretic and pain killer.
- Methyl salicylate = Oil of wintergreen (from methanol + salicylic acid).
Concepts [26]
- Concepts of Aldehydes, Ketones, and Carboxylic Acids
- Classification of Ketones
- Nature of Carbonyl Group
- Nomenclature of Aldehydes
- Nomenclature of Ketones
- Preparation of Aldehydes
- Preparation of Ketones
- Physical Properties of Aldehydes
- Physical Properties of Ketones
- Chemical Reactions of Aldehydes and Ketones - Nucleophilic Addition Reactions
- Chemical Reactions of Aldehydes and Ketones - Reduction
- Chemical Reactions of Aldehydes and Ketones - Oxidation
- Chemical Reactions of Aldehydes and Ketones - Reactions Due to α-hydrogen
- Chemical Reactions of Aldehydes and Ketones - Other Reactions
- Uses of Aldehydes and Ketones
- Carboxylic Acids
- Classification of Carboxylic Acids
- Nomenclature of Carboxylic Acids
- Structure of the Carboxyl group
- Methods of Preparation of Carboxylic Acids
- Physical Properties of Carboxylic Acids
- Chemical Reactions of Carboxylic Acids - Reactions Involving Cleavege of O-H Bond
- Chemical Reactions of Carboxylic Acids - Reactions Involving Cleavege of C-OH Bond
- Chemical Reactions of Carboxylic Acids - Reactions Involving –COOH Group
- Chemical Reactions of Carboxylic Acids - Substitution Reactions in the Hydrocarbon Part
- Uses of Carboxylic Acids
