Advertisements
Advertisements
प्रश्न
You are provided with a mixture of naphthalene and ammonium chloride by your teacher. Suggest an activity to separate them with well labelled diagram.
Advertisements
उत्तर
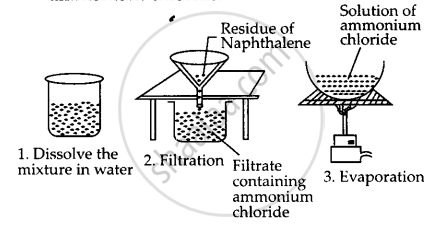
A mixture of naphthalene and ammonium chloride can be separated as follows :
Step-1: Put the mixture in a beaker and add water to it. Stir with a glass rod. Ammonium chloride is soluble in water gets dissolved leaving behind the insoluble naphthalene.
Step-2: Filter the solution. Naphthalene remains on the filter paper while ammonium chloride is obtained as filtrate.
Step-3: Evaporate the filtrate to get back ammonium chloride.
APPEARS IN
संबंधित प्रश्न
What are fluids ? Give two examples.
Write whether the following statement is true or false.
If the container in which a gas is collected has an opening, the gas will flow out and spread itself indefinitely.
Write true or false for statement. Rewrite the false statement correctly.
Solids and liquids can flow.
A kind of matter which can sublinje is
- water
- plastic
- milk
- iodine
Fill in the blank
The molecules are at a greater distance in ______ compared to liquids.
Fill in the blank:
The three states of matter are classified on the basis of differences of certain _________.
200 ml of water is poured into a bowl of 400 ml capacity. The volume of water will be ______.
Water obtained from tube wells is usually ______ water.
Mention any two characteristics of the particles of matter.
The property to flow is unique to fluids. Which one of the following statements is correct?
