Advertisements
Advertisements
प्रश्न
Write the structure of the product formed when chlorobenzene is treated with sodium metal in the presence of dry ether.
Advertisements
उत्तर
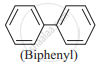
Biphenyl
Explanation:
Chlorobenzene + Sodium + Dry Ether → Biphenyl + NaCl
\[\ce{2C6H5Cl + 2Na \overset{dry ether}{->} C6H5 - C6H5 + 2NaCl}\]
APPEARS IN
संबंधित प्रश्न
Give reason:
Reactions involving Grignard reagent must be carried out under anhydrous condition.
Explain reactions of haloarenes with sodium metal.
The reaction in which 2 molecules of chlorobenzene reacts with metallic sodium in presence of dry ether forming diphenyl is an example of ______.
Identify the compound that gives 2, 5-dimethylhexane by reacting with sodium metal in presence of dry ether.
What is the action of following on ethyl bromide:
Na in dry ether
What is the action of following on ethyl bromide?
\[\ce{Mg}\] in dry ether
Write chemical reactions for the following conversions:
Bromobenzene to toluene.
The formula for the Grignard's reagent is ______.
Which of the following is called Wilkinson's catalyst?
The major product obtained in the following reaction is
\[\ce{Chlorobenzene + Chlorine ->[Anhydrous][FeCl3] product (Major)}\]
Identify electron withdrawing group from following when attached to π - bond.
Write the chemical reaction for the following:
Chlorobenzene is heated with fuming H2SO4.
From Wurtz reaction one can not produce:
Write the product formed during Chlorobenzene treated with chlorine reactions.
Write the product formed by chlorobenzene treated with a fuming H2SO4 reaction.
Write the product formed Anisole treated with HI reaction.
Define organometallic compound.
Write the product in the following reaction:
Bromobenzene reacts with methyl bromide and sodium metal in presence of dry ether.
Write the product in the following reaction:
Chlorobenzene reacts with sodium metal in presence of dry ether.
Identify the product 'Z' in the following series of reactions.
\[\ce{\underset{}{Ethanol} ->[SOCl2][Δ] \underset{\underset{}{}}{X}->[Mg][Dry ether] \underset{\underset{}{}}{Y}->[NH3] \underset{\underset{}{}}{Z}}\]
Identify the product ‘C’ formed in the following series of reactions.
\[\ce{Bromoethane ->[Mg][Dry ether] A ->[HOH][Dry ether] B ->[Br2][UV light] C}\]
