Advertisements
Advertisements
प्रश्न
Write Lewis symbols for the following atoms and ions: Al and Al3+.
Advertisements
उत्तर १
Al and Al3+
The number of valence electrons in aluminium is 3.
The Lewis dot symbol of aluminium (Al) is 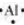 .
.
The tripositive charge on a species infers that it has donated its three electrons. Hence, the Lewis dot symbol is 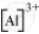
उत्तर २
`""_13"Al"` = 2,8,3 ∴ Lewis symbol = 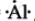 , `"Al"^(3+) "ions" = ["Al"]^(3+)`.
, `"Al"^(3+) "ions" = ["Al"]^(3+)`.
APPEARS IN
संबंधित प्रश्न
Write Lewis symbols for the following atoms and ions: S and S2–.
Write Lewis symbols for the following atoms and ions H and H–.
Use Lewis symbols to show electron transfer between the following atoms to form cations and anions:-
K and S
Draw Lewis dot diagram for the following.
Hydrogen (H2)
Draw Lewis dot diagram for the following.
Water (H2O)
Draw Lewis dot diagram for the following.
Methane (CH4)
Draw Lewis electron dot structure of C2H6
Draw Lewis electron dot structure of SO2
Explain in brief with one example of a covalent bond
Explain in brief with one example of coordinate bond
Which of the following molecules has a central atom with complete octet?
Which of the following is INCORRECT with respect to enthalpy?
Which of the following molecule does not obey octet rule?
What is the formal charge on hydrogen atom in water molecule?
Which of the following molecule does not obey octet rule?
Assertion (A): Sodium chloride formed by the action of chlorine gas on sodium metal is a stable compound.
Reason (R): This is because sodium and chloride ions acquire octet in sodium chloride formation.
What is the formal charge present on the oxygen atom (numbered 1) in the Lewis structure of CO2?
\[\ce{^\bullet_\bullet {\ce{O}} ≡ C - \overset{\bullet\bullet}{\underset{\bullet\bullet}{O^\bullet_\bullet}}}\]
