Advertisements
Advertisements
प्रश्न
Write the chemical equation for preparing sodium soap from glyceryl oleate . Structural formulae of the compounds are given below.
(C17H32COO)3C3H5 – Glyceryl oleate
Advertisements
उत्तर
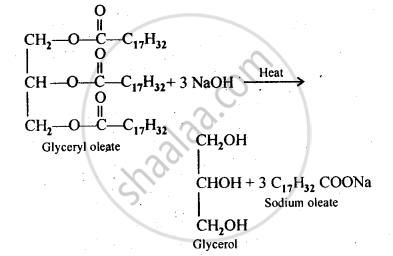
shaalaa.com
या प्रश्नात किंवा उत्तरात काही त्रुटी आहे का?
