Advertisements
Advertisements
प्रश्न
Why does the liquid rise in a syringe when its piston is pulled up?
Advertisements
उत्तर
When syringe is kept with its opening just inside a liquid and its plunger is pulled up in the barrel, the pressure of air inside the barrel below the plunger becomes much less than the atmospheric pressure acting on the liquid. As a result, the atmospheric pressure forces the liquid to rise up in the syringe.
APPEARS IN
संबंधित प्रश्न
You want to buy a house. Would you like to buy a house having windows but no ventilators? Explain your answer.
How does the pressure at a certain depth in sea water differ from that at the same depth in river water? Explain your answer.
What is the purpose of a barometer?
What is a barometer?
State two factors which affect the atmospheric pressure as we go up.
What will be the effect on air pressure if the temperature drops? Why?
The value of the atmospheric pressure at sea level is
What is meant by atmospheric pressure?
Figure shows a child blowing air with a straw near the opening of another straw which has its other end in a soft drink bottle. It was observed that the level of the soft drink in the straw rises up as soon as air is blown over its open end. Which one of the following best explains the reason for rise in level of the drink?
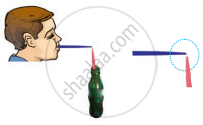
What is referred to as atmospheric pressure?
