Advertisements
Advertisements
प्रश्न
What is Grignard reagent? How is it prepared? Why are they prepared under anhydrous condition?
Advertisements
उत्तर
- Grignard reagent: When alkyl halide is treated with magnesium in the dry ether as the solvent, it gives alkyl magnesium halide. This is known as the Grignard reagent.
- The Grignard reagent can be prepared as,
\[\ce{\underset{Alkyl halide}{R - X} + Mg ->[dry ether] \underset{\text{Alkyl magnesium halide (Grignard reagent)}}{R - Mg - X}}\] - Grignard reagents are highly reactive compounds.
- They react with water or compounds containing hydrogen attached to the electronegative element.
Hence, reactions involving Grignard reagent must be carried out under anhydrous condition.
APPEARS IN
संबंधित प्रश्न
Write the structure and IUPAC name of the major product in the following reaction.

Write the structure and IUPAC name of the major product in the following reaction.
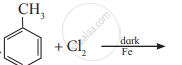
Convert the following:
But-1-ene to n-butyl iodide
Give reason. Though alkyl halides are moderately polar, they are insoluble in water.
Which of the following reaction proves the chlorinating property of phosphorus pentachloride?
Which of the following pairs of aryl halides cannot be prepared directly by electrophilic substitution?
The compound formed when 3, 5-dinitrobenzoic acid reacts with thionyl chloride is ___________.
Identify Sandmeyer's reaction from the following.
Alkyl halides can be hydrolysed to alcohols by reacting with ____________.
Amongst the following, HBr reacts fastest with
Which of the following alkyl halides has the lowest boiling point?
What is the relative rate of SN2 reaction for (CH3)3C - Br?
The reaction \[\ce{2R - Cl + COF2 -> 2R - F + COCl2}\] is an example of ______.
What is molecular formula of allyl chloride?
An alkyl iodide on refluxing with aqueous KOH solution gave isopropyl alcohol. The structure of alkyl iodide could be:
The conversion of ethyl bromide using sodium iodide and dry acetone, this reaction is know as ______.
Choose the false statement from the following.
Explain Swartz reaction.
Explain the preparation of alkyl halide using thionyl chloride (SOCl2).
What is the advantage of alkyl halide using the thionyl chloride (SOCl2 ) method?
Complete the following reaction giving major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{.................}\\
|\phantom{...................}\\
\ce{CH3 - C - CH2 - Cl ->[Na/dry ether] A}\\
|\phantom{...................}\\
\ce{CH3}\phantom{................}
\end{array}\]
Which of the following acid strengths is used to get a good yield of alkyl iodide from alcohol and sodium iodide in phosphoric acid?
