Advertisements
Advertisements
प्रश्न
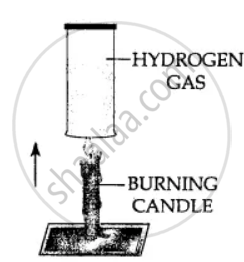
Using a burning candle and a jar of hydrogen – how would you prove experimentally that Hydrogen is a combustible gas.
Advertisements
उत्तर
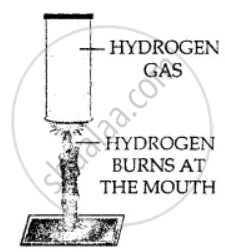
Hydrogen gas is combustible and bums at the mouth of the gas jar. When a burning candle is brought near the mouth of hydrogen in gas jar.
APPEARS IN
संबंधित प्रश्न
Indicate which of the following statement is true and which is false:
Hydrogen gas is easily liquefiable.
Give reason for the following:
A pop sound produced when hydrogen is burnt?
Define catalytic hydrogenation.
FILL IN THE BLANK
In nature, hydrogen occurs as a .................. molecule represented as H2
FILL IN THE BLANK
The compounds of carbon and hydrogen are called .................
Write the balanced equation and give your observation when the following metal reacts:
Calcium with cold water
Write the balanced equation and give your observation when the following metal reacts:
Magnesium with steam
Give the balanced equation of the reaction.
Give balanced equation for the following conversion:
Nitrogen to a basic gas – using hydrogen.
Write a correctly balanced equation for the following:
When steam is passed over red hot iron.
