Advertisements
Advertisements
प्रश्न
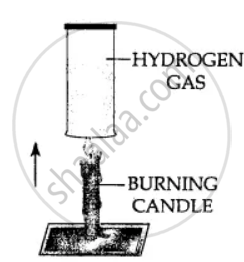
Using a burning candle and a jar of hydrogen – how would you prove experimentally that Hydrogen is a combustible gas.
Advertisements
उत्तर
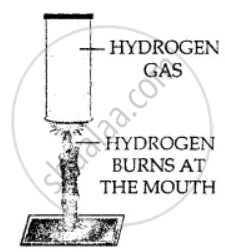
Hydrogen gas is combustible and bums at the mouth of the gas jar. When a burning candle is brought near the mouth of hydrogen in gas jar.
APPEARS IN
संबंधित प्रश्न
Give reason for the following:
A pop sound produced when hydrogen is burnt?
Give reason for the following:
Helium replaced hydrogen in weather observation balloons?
Give a balanced chemical equation for the reaction.
Multiple Choice Question
Hydrogen is responsible for producing
Why is hydrogen called a reducing agent? Give chemical equation to support your answer ?
Give equations for the reactions.
Give balanced equation for the following conversion:
Iron [III] oxide to iron – using hydrogen.
Give reason for the following:
The reaction of chlorine with hydrogen sulphide is deemed a redox reaction.
Match the statements in List I with the appropriate answer in List II.
| List I | List II |
| 1. An atom of hydrogen | A: Chromic oxide |
| 2. A strong oxidising agent | B: Ammonia |
| 3. A promoter used in Bosch process | C: Iron |
| 4. A chemical used in the manufavture of fertilizers | D: One electron |
| 5. The catalyst used in production of a basic gas from nitrogen. | E: Dilute nitric acid |
What do the following symbols [or formula] denote : 2H ; H2 ; H+. [two atoms, molecule, ion]
