Advertisements
Advertisements
प्रश्न

The X is
पर्याय
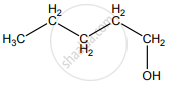
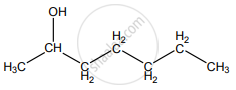
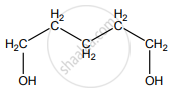
None of these
Advertisements
उत्तर

Explanation:
Hydro boration – Anti markownikoff product i.e CH3 – CH2 – CH – CH2 – CH2 – OH
APPEARS IN
संबंधित प्रश्न
Carbolic acid is ____________.
Isopropyl benzene on air oxidation in the presence of dilute acid gives ___________.
The reaction is an example of
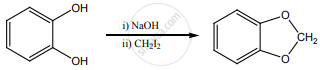
Draw the major product formed when 1-ethoxyprop-1-ene is heated with one equivalent of HI.
Arrange the following in the increasing order of their boiling point and give a reason for your ordering.
Butan-2-ol, Butan-1-ol, 2-methyl propane-2-ol
How is the conversion effected benzyl alcohol to benzoic acid?
0.44 g of a monohydric alcohol, when added to methyl magnesium iodide in ether, liberates at STP 112 cm3 of methane with PCC the same alcohol form a carbonyl compound that answers the silver mirror test. Identify the compound.
What will be the product (X and A) for the following reaction:
\[\ce{acetylchloride ->[i) CH3MgBr][ii) H3O+] X ->[acid K2Cr2O7]A}\]
What will be the product (X and A)for the following reaction
\[\ce{acetylchloride ->[i) CH3MgBr][ii) H3O] X ->[acid K2Cr2O7] A}\]
What will be the product (X and A) for the following reaction.
\[\ce{acetylchloride ->[i) CH3MgBr][ii) H3O^+] X ->[acid K2Cr2O7] A}\]
