Advertisements
Advertisements
प्रश्न
State Kelvin-Planck's statement of the second law of thermodynamics.
Advertisements
उत्तर
Kelvin-Planck's statement: It is impossible to construct a heat engine that operates in a cycle, whose sole effect is to convert the heat completely into work. This implies that no heat engine in the universe can have 100% efficiency.
APPEARS IN
संबंधित प्रश्न
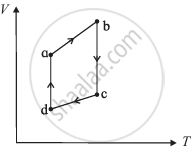
The figure shows the V-T diagram for one cycle of a hypothetical heat engine which uses the ideal gas. Draw the p-V diagram diagram of the system.
The efficiency of a heat engine working between the freezing point and boiling point of water is ____________.
State Clausius form of the second law of thermodynamics.
Explain the heat engine and obtain its efficiency.
Explain in detail the Carnot heat engine.
For a heat engine operating between temperatures t1 °C and t2 °C, its efficiency will be ______.
Heat engine transfers ______.
Let η1 is the efficiency of an engine at T1 = 447°C and T2 = 147°C while η2, is the efficiency at T1 = 947°C and T2 = 47°C. The ratio `eta_1/eta_2` will be ______.
The thermal efficiency of a heat engine is 25%. If in one cycle the heat absorbed from the hot reservoir is 50000 J, what is the heat rejected to the cold reservoir in one cycle?
What does a heat engine consist of?
