Advertisements
Advertisements
प्रश्न
Show that radius (rn) of the nth Bohr orbit varies directly with square of the principal quantum number (n) of the orbit.
Advertisements
उत्तर
Let r be the orbit’s radius, and e, m, and v represent the electron's charge, mass, and velocity, respectively. Z is the atomic number, and the nucleus has a positive charge of +Ze.
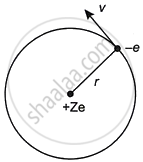
Centripetal force is provided by the electrostatic force of attraction.
Thus,
`(mv^2)/r = 1/(4 pi epsilon_0)`
or mv2 = `(Ze^2)/(4 pi epsilon_0 r)` ...(1)
From Bohr’s quantisation condition, we have oi
mvr = `n h/(2 pi)` ...(2)
n = 1, 2, 3, .... is the principal quantum number.
Squaring equation (2) and dividing by equation (1), we get
`(m^2v^2r^2)/(mv^2) = (n^2h^2//4 pi^2)/(Ze^2//4 pi epsilon_0 r)`
⇒ r = `(n^2h^2epsilon_0)/(pi m Ze^2)`
Since h, ε0, m, Z, and e are constants for an element.
Thus, rn ∝ n2
