Advertisements
Advertisements
प्रश्न
Out of C and CO, which is a better reducing agent for ZnO?
Advertisements
उत्तर १
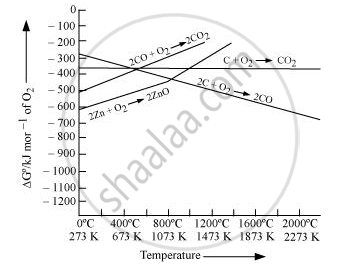
Reduction of ZnO to Zn is usually carried out at 1673 K. From the above figure, it can be observed that above 1073 K, the Gibbs free energy of formation of CO from C and above 1273 K, the Gibbs free energy of formation of CO2 from C is less than the Gibbs free energy of formation of ZnO. Therefore, C can easily reduce ZnO to Zn.
On the other hand, the Gibbs free energy of formation of CO2 from CO is always higher than the Gibbs free energy of formation of ZnO. Therefore, CO cannot reduce ZnO. Hence, C is a better reducing agent than CO for reducing ZnO.
उत्तर २
The two reduction reactions are:
\[\ce{ZnO_{(s)} + C_{(s)} -> Zn_{(s)} + CO_{(g)}}\] ...(i)
\[\ce{ZnO_{(s)} + CO_{(g)} -> Zn_{(s)} + CO2_{(g)}}\] ...(ii)
In the first case, there is an increase in the magnitude of ΔS° while in the second case, it almost remains the same. In other words, ΔG° will have a more negative value in the first case when C(s) is used as the reducing agent than in the second case when CO(g) acts as the reducing agent. Therefore, C(s) is a better reducing agent.
उत्तर ३
The C, CO curve in the Ellingham diagram is lower than the Zn, ZnO curve above 1120 K, while the C, CO2 curve is lower above 1323 K. As a result, at 1120 K and 1323 K, respectively, the ΔfG° of ZnO from C is higher than that of CO from C and CO2 from C. However, even beyond 2273 K, the CO, CO2 curve is higher than the Zn, ZnO curve. As a result, C can decrease ZnO, whereas CO cannot. Therefore, C is a better reducing agent for ZnO than CO.
संबंधित प्रश्न
Giving examples, differentiate between ‘roasting’ and ‘calcination’.
The metal oxide which cannot be reduced to metal by carbon is ____________.
Which one of the following is not feasible
Flux is a substance which is used to convert
Zinc is obtained from ZnO by ____________.
What are the various steps involved in the extraction of pure metals from their ores?
What is the role of Limestone in the extraction of iron from its oxide Fe2O3?
Explain the following term with a suitable example.
Gangue
Explain the following term with a suitable example.
Slag
Which reagents are required for one step conversion of chlorobenzene to toluene?
Extraction of gold and silver involves leaching the metal with CN– ion. The metal is recovered by ______.
At the temperature corresponding to which of the points in figure, FeO will be reduced to Fe by coupling the reaction \[\ce{2FeO -> 2Fe + O2}\] with all of the following reactions?
(a) \[\ce{C + O2 -> CO2}\]
(b) \[\ce{2C + O2 -> 2CO}\]
(c) \[\ce{2CO + O2 -> 2CO2}\]
(i) Point A
(ii) Point B
(iii) Point D
(iv) Point E
Colemanite is:-
Sulphide ore of the metal are usually concentrated by froth floatation process. Which of the following sulphide ore offers an exception and is concentrated by chemical leaching?
Heating byrites to remove sulphtir is called
Heating pyrites to remove sulphur is called ______.
