Advertisements
Advertisements
प्रश्न
Name the alkene which will yield 1-chloro-1-methylcyclohexane by its reaction with \[\ce{HCl}\]. Write the reactions involved.
Advertisements
उत्तर
Two alkenes are possible. These are methylene cyclohexane and 1 -Methylcyclohex-1-ene
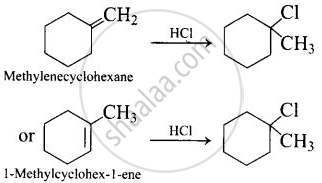
APPEARS IN
संबंधित प्रश्न
Write the formula of the following compound:
Potassiumhexacyanoferrate(III)
B.H.C. is used as an ____________.
Among the following, insecticide is:
Chloropicrin is formed by the reaction of ____________.
\[\ce{CH3CH2CH2Br + NaCN -> CH3CH2CH2CN + NaBr}\], will be fastest in:
A dihalogen derivative ‘X’ of a hydrocarbon with three carbon atoms react with ale. KOH and produces hydrocarbon which forms red ppt. with ammonical Cu2Cl2. ‘X’ gives an aldehyde on reaction with aq. KOH. The compound ‘X’ is:
Major product obtained on reaction of 3-Phenyl propene with HBr in presence of organic peroxide.
What are the IUPAC names of the insecticide DDT and benzene hexachloride? Why is their use banned in India and other countries?
Which of the following does not give iodoform test?
Chlorination of CS2 give
Chloroform on reduction with Zn an HCI (all) gives:-
The compound that will not give iodoform on treatment with alkali and iodine is ______.
The major product formed when 1, 1, 1-trichloropropane is treated with aqueous potassium hydroxide, is ______.
Auto-oxidation of chloroform in air and sunlight produces a poisonous gas known as ______.
Give the IUPAC names of the following componus:
\[\ce{CICH2C =CCH2Br}\]
Write equation to convert Methyl cyanide to ethanoic acid.
DDT is obtained by the reaction of chlorobenzene with ______.
Methyl bromide is not used ______.
