Advertisements
Advertisements
प्रश्न
In the cubic close packing, the unit cell has ______.
पर्याय
4 tetrahedral voids each of which is shared by four adjacent unit cells.
4 tetrahedral voids within the unit cell.
8 tetrahedral voids each of the which is shared by four adjacent unit cells.
8 tetrahedral voids within the unit cells.
Advertisements
उत्तर
In the cubic close packing, the unit cell has 8 tetrahedral voids within the unit cells.
Explanation:
Eight tetrahedral voids per fee unit cell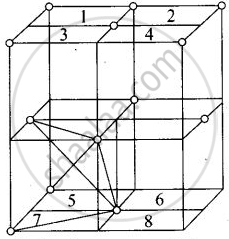
Each cube represented by numeric 1, 2, 3, 4, 5, 6, 7, 8 contains one tetrahedral void.
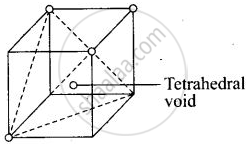
Each cube contains one tetrahedral void at its body centre as shown above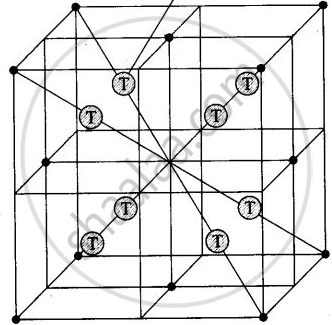
APPEARS IN
संबंधित प्रश्न
How will you distinguish between the following pair of terms?
Tetrahedral and octahedral voids
If the radius of the octahedral void is r and the radius of the atoms in close packing is R, derive the relation between r and R.
The unit cell of a substance has cations A+ at the corners of the unit cell and the anions B− at the center. The simplest formula of the substance is ____________.
Total number of voids in 0.5 mole of a compound which forms hexagonal close packed structure is ____________.
A compound is formed by two elements A and B. The element B forms cubic close packing and atoms of A occupy `1/3"rd"` of tetrahedral voids. The formula of this compound is ____________.
The Ca2+ and F– are located in CaF2 crystal, respectively at face centered cubic lattice points and in ____________.
Which set of following characteristics for ZnS crystal is correct?
Percentages of free space in cubic close packed structure and in the body-centered packed structure are respectively:
In which of the following crystals alternate tetrahedral voids are occupied?
In the hexagonal close-packed structure of a metallic lattice, the number of nearest neighbours of a metallic atom is ____________.
Which of the following is not true about the voids formed in 3 dimensional hexagonal close packed structure?
(i) A tetrahedral void is formed when a sphere of the second layer is present above triangular void in the first layer.
(ii) All the triangular voids are not covered by the spheres of the second layer.
(iii) Tetrahedral voids are formed when the triangular voids in the second layer lie above the triangular voids in the first layer and the triangular shapes of these voids do not overlap.
(iv) Octahedral voids are formed when the triangular voids in the second layer exactly overlap with similar voids in the first layer.
The number of tetrahedral voids per unit cell in NaCl crystal is:
(i) 4
(ii) 8
(iii) twice the number of octahedral voids.
(iv) four times the number of octahedral voids.
In a compound, nitrogen atoms (N) make cubic close packed lattice and metal atoms (M) occupy one-third of the tetrahedral voids present. Determine the formula of the compound formed by M and N?
A solid compound XY has Nacl structure. If the radium of cation (X+) is 100 pm, the radium of anion (r–) will be:-
A compound is formed by two elements A and B. The element B forms a cubic close-packed structure and atoms of A occupy `1/3` of the tetrahedral voids. If the formula of the compound is AxBy then the value x + y is in the option:
