Advertisements
Advertisements
प्रश्न
If 4.5kg of fuel is completely burnt and amount of heat produced stands measured at 1, 80,000 KJ what is the calorific value.
Advertisements
उत्तर
Calorific value = `"Heat Produced"/"Amount of Fuel"="1,80,000"/"4.5"= 40000`
∴ Calorific value = 40,000 kj/kg
APPEARS IN
संबंधित प्रश्न
Which is the hottest part in the flame of candle ______.
True or False – If False give the correct answer
All fuels form flame.
Inner zone of flame : :______, outer zone of flame : :______
Make labeled diagram of a candle flame.
Picture based question
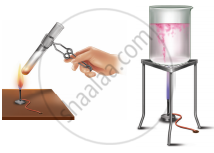
Arul and Aakash were doing an experiment in which water was to be heated in a beaker. Arul kept the beaker near the wick in the yellow part of candle flame. Aakash kept the beaker in the outer most part to the flame. Whose water will get heated in a shorter time?
The ______ temperature at which a substance catches fire is called its ______ temperature.
Write True/False against the following statement and also correct the false statement.
The substances which vapourise during burning, give flame.
Which is the luminous part of the flame?
Name a few substances which produce flames on burning.
Why is the candle flame straight?
