Advertisements
Advertisements
प्रश्न
If 4.5kg of fuel is completely burnt and amount of heat produced stands measured at 1, 80,000 KJ what is the calorific value.
Advertisements
उत्तर
Calorific value = `"Heat Produced"/"Amount of Fuel"="1,80,000"/"4.5"= 40000`
∴ Calorific value = 40,000 kj/kg
APPEARS IN
संबंधित प्रश्न
Abida and Ramesh were doing an experiment in which water was to be heated in a beaker. Abida kept the beaker near the wick in the yellow part of the candle flame. Ramesh kept the beaker in the outermost part of the flame. Whose water will get heated in a shorter time?
Which is the hottest part in the flame of candle ______.
Picture based question
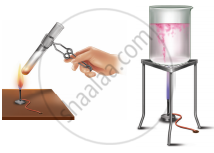
Arul and Aakash were doing an experiment in which water was to be heated in a beaker. Arul kept the beaker near the wick in the yellow part of candle flame. Aakash kept the beaker in the outer most part to the flame. Whose water will get heated in a shorter time?
On placing an inverted tumbler over a burning candle, the flame extinguishes after some time. This is because of non-availability of
The substances which vaporise during ______ give flame.
Write True/False against the following statement and also correct the false statement.
Middle zone is the hottest zone of a flame.
You are provided with three watch glasses containing milk, petrol and mustard oil respectively. Suppose you bring a burning candle near these materials one by one, which material(s) will catch fire instantly and why?
Match the following:
| 1. | White flame | a. | Table salt |
| 2. | Indigo flame | b. | Bleaching powder |
| 3. | Blue flame | c. | Potassium chloride |
| 4. | Orange flame | d. | Epsom salt |
Name a few substances which produce flames on burning.
Explain the structure of a candle flame.
