Advertisements
Advertisements
प्रश्न
How propanal is converted into propane.
Advertisements
उत्तर
\[\ce{\underset{(Propanal)}{CH3 - CH2} - CHO + 4[H] ->[Zn-Hg, conc.HCl][Δ] \underset{(Propane)}{CH3 - CH2} - CH3 + H2O}\]
APPEARS IN
संबंधित प्रश्न
Answer in brief.
What is the action of hydrazine on cyclopentanone in presence of KOH in ethylene glycol?
Identify A in the following reaction.
\[\ce{A + CH3MgBr ->[Ether] complex ->[H3O+] (CH3)3C - OH}\]
Which of the following gives red precipitate with Fehling's solution?
Which of the following represents the structure of isopropyl methyl ketone?
Which of the following does not give yellow solid on treatment with sodium hypoiodlte?
Acetone on Clemmensen reduction gives ____________.
Which of the following is a Wolf - Kishner reduction?
The formation of cyanohydrin from acetone is an example of ____________.
Assertion: p-N, N-dimethyl amino benzaldehyde undergoes benzoin condensation
Reason: The aldehydic (−CHO) group is meta directing.
Phenyl methanal is reacted with concentrated NaOH to give two products X and Y. X reacts with metallic sodium to liberate hydrogen X and Y are ____________.
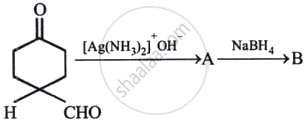
Identify A, B and C.

How will you convert benzaldehyde into the following compound?
Benzophenone
How will you convert benzaldehyde into the following compound?
α-hydroxy phenyl acetic acid
Write the structure of the major product of the aldol condensation of benzaldehyde with acetone.
How is the following conversion effected propanal into butanone?
How is the following conversion effected Hex-3-yne into hexan-3-one?
How is the following conversion effected phenyl methanal into benzoic acid?
How will you prepare cinnamic acid from benzaldehyde?
Which of the following compounds is most reactive towards nucleophilic addition reactions?
Which of the following compound will show a positive silver mirror test?
Which of the following compounds will undergo self-aldol condensation in the presence of cold dilute alkali?
Which of the following reaction does not involve either oxidation or reduction?
Which of the following is most reactive towards the addition reaction of hydrogen cyanide to form the corresponding cyanohydrin?
Which of the following compounds does not undergo a haloform reaction?
Which of the following is not correctly matched?
Write the structure of the major product of the aldol condensation of benzaldehyde with acetone.
Write the structure of the major product of the aldol condensation of benzaldehyde with acetone.
The products formed in the following reaction, A and B are:
Which from following compounds does NOT undergo haloform reaction?
