Advertisements
Advertisements
प्रश्न
How is the gas collected and why?
Advertisements
उत्तर
The gas is collected by upward displacement of air because -
(i) It is heavier than air.
(ii) It is highly soluble in water.
APPEARS IN
संबंधित प्रश्न
The aim of the Fountain experiment is to prove that ______.
Study the figure given below and answer the questions which follow:
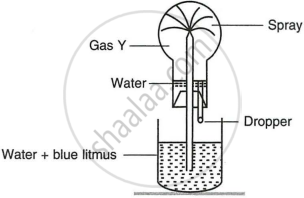
- Identify the gas Y.
- What property of gas Y does this experiment demonstrate?
- Name another gas which has the same property and can be demonstrated through this experiment.
Name :
Two colourless gases which when mixed produce a white solid.
Name the drying agent used to dry HCl gas.
Describe an experiment to prove the following:
HCI gas is heavier than air.
Give reason for the following:
Dilute hydrochloric acid cannot be concentrated by distilling (boiling) the dilute acid.

(a) Name the experiment illustrate above.
(b) Which property of hydrogen chloride is demonstrated by this experiment?
(c) State the colour of the water that has entered the round-bottomed flask.

The diagram shows an apparatus for the laboratory preparation of hydrogen chloride.
(i) Identify A and B
(ii) Write the equation for the reaction
(iii) How would you check whether or not the gas jar is filled with hydrogen chloride?
(iv) What does the method of collection tell you about the density of hydrogen chloride.
The aim of the fountain experiment is to prove that
State the following:
The drying agent used in the laboratory preparation of HCl gas.
