Advertisements
Advertisements
प्रश्न
Give reasons why – separation of mixtures e.g. iron from a mixture of iron and sulphur is a physical change, but heating a mixture of iron and sulphur is considered a chemical change.
Advertisements
उत्तर
Physical change
- There is no change in the specific properties of the constituents.
Example: The particles of iron can be separated from sulphur with the help of a magnet or by dissolving sulphur in carbon disulphide. - No new substance is formed.
Example: The mixture of iron and sulphur does not form any new substance. - There is no net release or absorption of energy.
Example: When iron and sulphur particles are mixed, heat energy is neither given out nor absorbed. - It is a temporary change and can be reversed by removing the cause of the change.
Example: The particles of iron can be separated from the mixture with the help of a magnet. removing the cause of change.
Chemical change
- There is a specific change in the properties of the constituents.
Example: The particles of iron or sulphur cannot be separated by a magnet or carbon disulphide. - A new substance is formed.
Example: Iron sulphide is the new substance formed when iron and sulphur are heated. - There is a net release or absorption of energy when a chemical change takes place.
Example: When iron sulphide is formed, the test tube gets red hot on account of the release of a large amount of heat energy. - It is a permanent change and cannot be reversed by removing the cause of the change.
Example: The particles of iron can be separated from the mixture with the help of a magnet. removing the cause of change.
APPEARS IN
संबंधित प्रश्न
Try segregating the things around you as pure substances or mixtures.
Fill in the blank.
Changes that occur in nature are called ........................
State four differences between physical and chemical changes.
State whether the following is physical or chemical changes.
cutting wood into small pieces —
Solve this crossword by using the clues that follow-
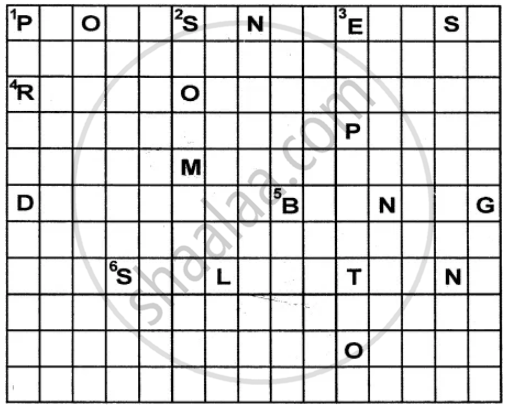
Across
1. Energy in the form of sunlight is absorbed by the green plants in this process.
4. The reddish-brown substance formed over iron in the presence of oxygen and moisture.
5. This change is permanent and irreversible.
6. The process in which a solid directly changes into gaseous state.
Down
1 . It is the change that takes place in case of swinging pendulum of a clock. –
2. Occurrence of this is a non-periodic change as well as a natural change.
3. It is a physical change.
Make a list of different types of changes with example.
What will you take into account while identifying the following?
A physical change in a substance.
State the observations seen, when milk in a dish is kept aside for a few hours or more. Is the change which occurs – a physical change or a chemical change. Give reasons.
Select the chemical changes from the following list of changes
Butter turning rancid
Adding sugar to milk is a physical change.
