Advertisements
Advertisements
प्रश्न
Explain in brief the involvement of energy in physical and chemical changes.
Advertisements
उत्तर
Involvement of energy in physical and chemical changes
- Physical changes use energy to change the state of matter,
- Chemical changes release or absorb energy when changing a substance into a new substance.
Energy is released during the decomposition of a substance while energy is absorbed during the formation of a new substance.
APPEARS IN
संबंधित प्रश्न
What do you observe when :
a spoon of sugar is heated in a pan
Tick (√) the correct alternative from the choice given for the following statement
When sugar is heated, its colour changes into
True or false?
During a physical change energy is either given out or absorbed by the system.
Under which different types can the following changes be classified?
Conversion of milk into yoghurt.
Fill in the blanks with the appropriate words of the statement given below:
Chemical changes may involve exchange of energy in the form of __________ and _________.
Picture based question
Observe the picture and list down the changes that are accompanied in the picture.

- ______.
- ______.
- ______.
Observe the picture containing a kettle and note that it has salt water in it and answer the following questions:
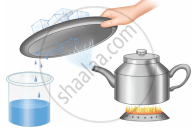
- What is name of the process that is done to the kettle?
- What will happen to the content of the kettle?
- What kind of change is occurring on the cold surface of the metal plate?
- What can you say about the quality of water that is obtained in the beaker?
Some changes are listed in the following table. For each change, write in the blank column, whether the change can be reversed or not.
| S. No. | Change | Can be reversed (Yes/No) |
| 1. | The sawing of a piece of wood | |
| 2. | The melting of ice candy | |
| 3. | Dissolving sugar in water | |
| 4. | The cooking of food | |
| 5. | The ripening of a mango | |
| 6. | Souring of milk |
______ is the process in which something becomes different from what it was earlier.
Cut vegetables turn brown when exposed to air, this is due to ______.
