Advertisements
Advertisements
प्रश्न
Give reason for the following:
When concentrated sulphuric acid is added to sugar/glucose, a black mass is left behind.
Advertisements
उत्तर
The carbohydrates contain carbon, hydrogen and oxygen. The hydrogen and oxygen are always in ratio of 2:1 which is absorbed by acid in the form of water, thus leaving carbon behind. This is also called as charring.
APPEARS IN
संबंधित प्रश्न
Name the products formed when hot and concentrated sulphuric acid reacts with Sugar.
Give reason for the following:
When concentrated sulphuric acid is exposed to air, its volume increases and it becomes slightly dilute.
How are the following conversion brought about? Give equation and condition:
Sucrose to sugar charcoal.
Give examples of the use of sulphuric acid as
An oxidizing agent
Sulphuric acid can be used to prepare a number of gases in the laboratory. Write balanced equation for the reaction from which the folllowing gas are obtained, using dilute sulphuric acid as one of the reactant : Sulphur dioxide
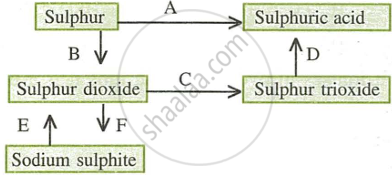
- Name the catalyst which helps in the conversion of sulphur dioxide to sulphur trioxide in step C.
- In the contact process for the manufacture of sulphuric acid, sulphur trioxide is not converted to sulphuric acid by reacting it with water. Instead a two-step procedure is used. Write the equations for the two steps involved in D.
- What type of substance will liberate sulphur dioxide from sodium sulphite in step E?
- Write the equation for the reaction by which sulphur dioxide is converted to sodium sulphite in step F.
Write the balanced chemical equation for the following conversion:
Ammonium sulphate from ammonia and dilute sulphuric acid.
Write a balanced equation for the following conversion:
Lead sulphate from lead nitrate and sulphuric acid.
Convert the following reaction into a balanced chemical equation:
Sodium hydroxide to sodium sulphate using sulphuric acid.
Hydrogen chloride gas is prepared in the laboratory by the action of concentrated sulphuric acid on sodium chloride.
State the method of collection of the gas formed above.
