Advertisements
Advertisements
प्रश्न
Give the name and formula of two : Weak dibasic acids
Advertisements
उत्तर
(i) Carbonic acid H2CO3
(ii) Oxalic acid (COOH)2
APPEARS IN
संबंधित प्रश्न
Ammonia is found in many household products, such as window cleaners. It turns red litmus blue. What is its nature?
When a solution is added to a cloth strip treated with onion extract, then the smell of onion cannot be detected. State whether the given solution contains an acid or a base.
Which gas is usually liberated when an acid reacts with a metal? How will you test for the presence of this gas?
Give the names and formulae of two strong bases and two weak bases.
The label on the bottle of chemical is spoiled. How will you find whether the chemical is acidic or not?
Which of the following is not a mineral acid?
Fill in the cross word given in figure with the help of the clues provided.
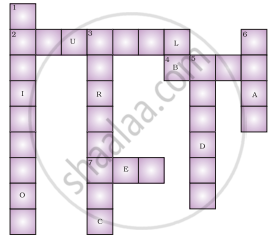
Across
(2) The solution which does not change the colour of either red or blue litmus.
(4) Phenolphthalein gives pink colour in this type of solution.
(7) Colour of blue litmus in lemon juice.
Down
(1) It is used to test whether a substance is acidic or basic.
(3) It is a natural indicator and gives pink colour in basic solution.
(5) Nature of ant’s sting.
(6) It is responsible for the increase in temperature during a neutralisation reaction.
Write the important uses of hydrochloric acid and sulphuric acids.
Which one of the following acids undergoes complete ionisation?
______ acids have a relatively smaller amount of acids dissolved in a solvent.
