Advertisements
Advertisements
प्रश्न
Give a balanced equation when dilute hydrochloric acid is added to : Magnesium bicarbonate
Advertisements
उत्तर
Mg(HCO3)2 + 2HCI → MgCl2 + 2H2O +2CO2
APPEARS IN
संबंधित प्रश्न
Hydrochloric acid contains (i) hydrogen (ii) chlorine. Prove it. Write equations for the reactions.
Name the experiment which demonstrates that hydrogen chloride is soluble in water.
Give reason for the following:
silver nitrate solution can be used to distinguish HCl from HNO3
Distinguish between the following pairs of compounds using the reagent given in the bracket.
Mangenese dioxide and copper (II) oxide. (using concentrated HCl)
How will you prove that hydrochloric acid contains
- hydrogen
- chlorine?
Write equations for the reactions.
Convert Hydrochloric acid to nascent chlorine.
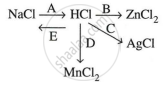
Study the flow chart and give balanced equations with conditions for the conversions A, B, C, D, and E.
Complete and balance the following reaction, state whether dilute or cone. acid is used.
\[\ce{NH4OH + HCl ->}\]
Convert Hydrochloric acid to nascent chlorine.
Complete and balance the following reaction, state whether dilute or cone. acid is used.
\[\ce{NH4OH + HCl->}\]
