Advertisements
Advertisements
प्रश्न
Give balanced chemical equation to prepare the following salt:
Lead sulphate from lead carbonate
Advertisements
उत्तर
\[\ce{PbCO3 + 2HNO3 -> Pb(NO3)2 + H2O + CO2}\]
\[\ce{Pb(NO3)2 + H2SO4 -> PbSO4 + 2HNO3}\]
APPEARS IN
संबंधित प्रश्न
Identify the acid in the following case:
The acid which produces sugar charcoal from sugar.
State the conditions required for the given reaction to take place:
Any two conditions for the conversion of sulphur dioxide to sulphur trioxide
Which property of sulphuric acid is shown by the reaction of the concentrated sulphuric acid with Ethanol?
Why is concentrated sulphuric acid kept in air tight bottles?
Give reason for the following:
When solution of sulphur dioxide is exposed to air, it gets converted to sulphuric acid.
Describe the reaction that show
Dilute sulphuric acid behaves as dibasic acid.
Why is sulphuric acid known as king of chemicals and oil vitriol ?
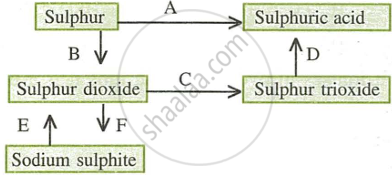
- Name the catalyst which helps in the conversion of sulphur dioxide to sulphur trioxide in step C.
- In the contact process for the manufacture of sulphuric acid, sulphur trioxide is not converted to sulphuric acid by reacting it with water. Instead a two-step procedure is used. Write the equations for the two steps involved in D.
- What type of substance will liberate sulphur dioxide from sodium sulphite in step E?
- Write the equation for the reaction by which sulphur dioxide is converted to sodium sulphite in step F.
State your observation for the following case:
Paper soaked in potassium permanganate solution is introduced into a gas jar of sulphur dioxide.
In the given equation identify the role played by concentrated sulphuric acid S + 2H2SO4→ 3SO2 + 2H2O
