Advertisements
Advertisements
प्रश्न
Following type of nom-ionic detergents are present in liquid detergents, emulsifying agents and wetting agents. Label the hydrophilic and hydrophobic parts in the molecule. Identify the functional group (s) present in the molecule.

Advertisements
उत्तर
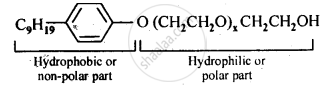
Functional groups present in the molecule are:
(i) Ether, and
(ii) primary alcoholic group
shaalaa.com
या प्रश्नात किंवा उत्तरात काही त्रुटी आहे का?
