Advertisements
Advertisements
प्रश्न
Find the atomicity of the Following molecule:
Dinitrogen Oxide
Advertisements
उत्तर
Atomicity of:
Dinitrogen Oxide - N2O
= 1 X 2 + 1 = 2 + 1 = 3
APPEARS IN
संबंधित प्रश्न
Name the technique that you use if only the solid component of a solution is required.
State the number of atoms of H2SO4
Name: Two substances used to make electric wires
Give reason: Argon is filled in electric bulbs.
Differentiate between the terms –
elements, compounds & mixtures.
The important physical properties of substances are colour, odour, nature, density & solubility in water.
Name – two coloured gases (with their colours)
Give the name of the radical - CO3
Explain the difference between Atoms and molecules.
Classify the following as metals, non-metals and metalloids.
Sodium, Bismuth, Silver, Nitrogen, Silicon, Carbon, Chlorine, Iron, Copper
Higher Order Thinking Skills.
In the diagram given below, the circle, square and triangle represent the atoms of different elements.
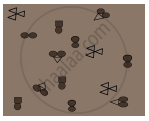
Identify all combinations that represent
- molecule of a compound
- molecule of an element consisting of two atoms
- molecule of an element consisting of three atoms
