Advertisements
Advertisements
प्रश्न
Explain how frequency of incident light varies with stopping potential.
Advertisements
उत्तर
1. The effect of frequency of incident light on stopping potential can be studied by keeping the intensity of the incident light constant.
2. The electrode potential varies for different frequencies of the incident light.
3. As the frequency is increased the photoelectrons are emitted with greater kinetic energies so that the retarding potential needed to stop the photoelectrons is also greater.
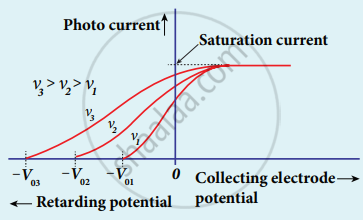
Variation of photocurrent with collector electrode potential for different frequencies of the incident radiation
4. From the graph between frequency and stopping potential, the stopping potential varies linearly with the frequency of the incident light.
5. The stopping potential is zero when no electrons are emitted below a certain frequency called threshold frequency.

Variation of stopping potential with frequency of the incident radiation for two metals
APPEARS IN
संबंधित प्रश्न
A photoelectric surface is illuminated successively by monochromatic light of wavelength λ and `λ/2`. If the maximum kinetic energy of the emitted photoelectrons in the second case is 3 times that in the first case, the work function of the material is
In photoelectric emission, a radiation whose frequency is 4 times threshold frequency of a certain metal is incident on the metal. Then the maximum possible velocity of the emitted electron will be
A light of wavelength 500 nm is incident on a sensitive metal plate of photoelectric work function 1.235 eV. The kinetic energy of the photoelectrons emitted is (Take h = 6.6 × 10–34 Js)
What is a surface barrier?
Mention the two features of x-ray spectra, not explained by classical electromagnetic theory.
Briefly discuss the observations of Hertz, Hallwachs and Lenard.
Explain the effect of potential difference on photoelectric current.
When a light of frequency 9 × 1014 Hz is incident on a metal surface, photoelectrons are emitted with a maximum speed of 8 × 105 ms−1. Determine the threshold frequency of the surface.
When a 6000 Å light falls on the cathode of a photo cell, photoemission takes place. If a potential of 0.8 V is required to stop emission of electron, then determine the
- frequency of the light
- energy of the incident photon
- work function of the cathode material
- threshold frequency and
- net energy of the electron after it leaves the surface.
UV light of wavelength 1800 Å is incident on a lithium surface whose threshold wavelength is 4965 Å. Determine the maximum energy of the electron emitted.
