Advertisements
Advertisements
प्रश्न
|
Conductivity of Aqueous Solutions:
Electrical conductivity is based on the flow of ions. Slightly ionized substances are weak electrolytes. Weak acids and bases would be categorized as weak electrolytes because they do not completely dissociate in solution. Highly ionized substances are strong electrolytes. Strong acids and salts are strong electrolytes because they completely ionize in solution. The ions carry the electric charge through the solution, thus creating an electric current. The current, if sufficient enough, will light one or both LEDs on a conductivity meter, shown at right. The meter has a 9V battery, two parallel copper electrodes and two LEDs, one green and one red. The conductivity of a solution can be tested by switching the meter on and dipping the copper electrodes in the solution to be tested. Substances that do not conduct an electric current are called non-electrolytes. Non-electrolytes do not ionize; they do not contain moveable ions. The LEDs of a conductivity meter will not light because there are no ions to carry the electric current. The table given below is a guide to the possible conductivity measurements.
|
Based on the information provided above, answer the following questions:
I. Is it possible to identify whether the given solution is 1 M NaOH or 1 M HCl using the conductivity meter? Justify your answer. (1)
II. What is the possible pH value of the solution if the glow of the green LED is medium and the red LED glows very brightly? (1)
(i) 1 (ii) 13 (iii) 5 (iv) 8
- (i) and (ii)
- (i) and (iii)
- (ii) and (iv)
- (iii) and (iv)
OR
Write down the observations if the conductivity meter is dipped in distilled water.
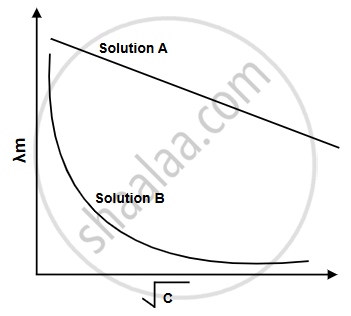
III. Refer to the table given below and draw the molar conductivity vs. concentration curve for solutions A and B. (2)
| Solution | Red LED | Green LED |
| A | bright | dim |
| B | dim | off |
Advertisements
उत्तर
I. No. Both have strong electrolytes, so both LEDs will glow. In both cases, red will be very bright, and green will be medium.
II. (i) and (ii)
OR
| Scale | Red LED | Green LED | Conductivity |
| 0 | off | off | low or none |
III. A is a strong electrolyte, while B is a weak electrolyte.