Advertisements
Advertisements
प्रश्न
Draw the number of scattered particles versus the scattering angle graph for scattering of alpha particles by a thin foil. Write two important conclusions that can be drawn from this plot.
आलेख
लघु उत्तर
Advertisements
उत्तर
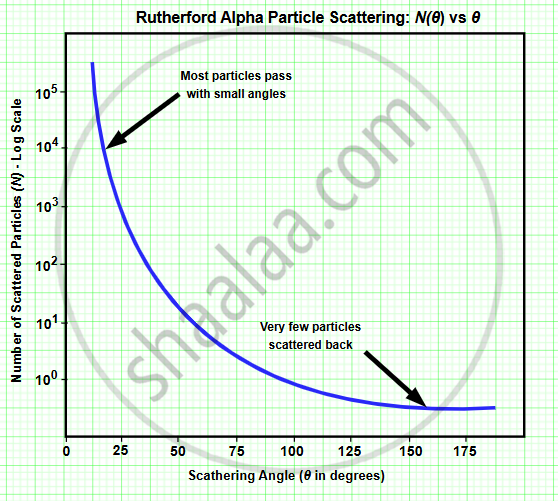
Conclusions:
- An atom is mostly empty space: Since most alpha particles pass through with little or no deflection, positive charge and mass are not uniformly spread, and most of the atom is empty.
- Presence of a small, dense nucleus: A very small fraction of particles are deflected through large angles, indicating a strong repulsive force and positive charge is concentrated in a tiny central region (nucleus).
shaalaa.com
या प्रश्नात किंवा उत्तरात काही त्रुटी आहे का?
2025-2026 (March) 55/5/1
