Advertisements
Advertisements
प्रश्न
Dilute NaOH can be tested with
(a) red litmus paper
(b) blue litmus paper
(c) lime water
(d) Na2CO3
Advertisements
उत्तर
(a) red litmus paper
Sodium hydroxide when dissolved in water undergoes the following
reaction:
NaOH(s) + aqs → Na+(aq) + OH−(aq)
The dissociated OH− ions turn moist red litmus paper blue.
APPEARS IN
संबंधित प्रश्न
How is the concentration of hydronium ions (H3O+) affected when the solution of an acid is diluted?
A solution reacts with crushed egg-shells to give a gas that turns lime-water milky. The solution contains ______.
Write a word equation and then a balanced equation for the reaction taking place when:
Dilute hydrochloric acid reacts with iron filings.
Give chemical equation of the reaction involved.
What happens when an acid reacts with a metal carbonate? Explain with the help of an example.
Compounds such as alcohol and glucose also contain hydrogen but are not categorised as acids. Describe an activity to prove it.
On adding dilute hydrochloric acid to copper oxide powder, the solution formed is blue-green.
On the basis of the above reaction, what can you say about the nature of copper oxide?
What ions are present in the solutions of following substances? (write the symbols only)
Sodium hydroxide
Hydrochloric acid reacts with a metal X to form a gas Y which burns with a 'pop' sound. Sodium hydroxide solution also reacts with the same metal X (on heating) to form the same gas Y.
Name X and Y
Name a metal compound which has detergent properties (cleansing properties).
What are esters? How are esters prepared? Write the chemical equation for the reaction involved. What happens when an ester reacts with sodium hydroxide? Write the chemical equation for the reaction and also state the name and use of this reaction.
The table given below shows the reaction of a few elements with acids and bases to evolve Hydrogen gas.
| Element | Acid | Base |
| A |
✗
|
✗ |
| B |
✓
|
✓ |
| C | ✓ | ✗ |
| D | ✓ | ✓ |
Which of these elements form amphoteric oxides?
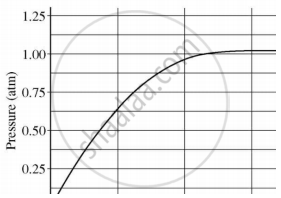
A student added 10 g of calcium carbonate in a rigid container, secured it tightly and started to heat it. After some time, an increase in pressure was observed, the pressure reading was then noted at intervals of 5 mins and plotted against time, in a graph as shown below. During which time interval did maximum decomposition take place?
If a few drops of a concentrated acid accidentally spills over the hand of a student, what should be done?
Which of the following statements is not correct?
Name the acid present in ant sting and give its chemical formula. Also give the common method to get relief from the discomfort caused by the ant sting.
Can we taste acids and bases to identify them?
Consider the following salt:
\[\ce{ZCO3}\]
What would be the change in colour in blue litmus if \[\ce{ZCO3}\] is added to it and Z is potassium?
