Advertisements
Advertisements
प्रश्न
Choose the correct answer from the options given below :
Hydroxide of this metal is soluble is sodium hydroxide solution
पर्याय
Magnesium
Lead
Silver
Copper
Advertisements
उत्तर
Lead
APPEARS IN
संबंधित प्रश्न
Name a yellow monoxide that dissolves in hot and concentrated caustic alkali.
Using sodium hydroxide solution, how will you distinguish:
Lead hydroxide from magnesium hydroxide.
Three test tubes contain calcium nitrate solution, zinc nitrate solution and lead nitrate solution.Describe the effect of Adding sodium hydroxide solution to each portion in turn till it is in excess.
Write the observation, and balanced equation for the following reaction:
Sodium hydroxide is added dropwise to a solution of zinc sulphate, till it is in excess.
Sodium hydroxide solution is added first in a small quantity, then in excess to the aqueous salt solutions of copper (II) sulphate, zinc nitrate, lead nitrate, calcium chloride, and iron (III) sulphate. Copy the following table and write the colour of the precipitate in (i) to (v) and the nature of the precipitate (soluble or insoluble) in (vi) to (x).
| Aqueous salt solution | Colour of precipitate when NaOH is added in a small quantity | Nature of precipitate (soluble or insoluble) when NaOH is added in excess |
| Copper (II) sulphate | (i) | (vi) |
| Zinc nitrate | (ii) | (vii) |
| Lead nitrate | (iii) | (viii) |
| Calcium chloride | (iv) | (ix) |
| Iron (III) Sulphate | (v) | (x) |
Distinguish by adding:
Sodium hydroxide solution and ammonium hydroxide solution to lead nitrate solution and zinc nitrate solution.
Distinguish by adding:
Sodium hydroxide solution and ammonium hydroxide solution to Fe(II) salt solution and Fe(III) salt solution.
Distinguish by adding:
Sodium hydroxide solution and ammonium hydroxide solution to ferrous nitrate and lead nitrate.
A student was asked to perform an experiment in the laboratory based on the instructions given:
Observe the picture given below and state one observation for the experiment you would notice on mixing the given solution.
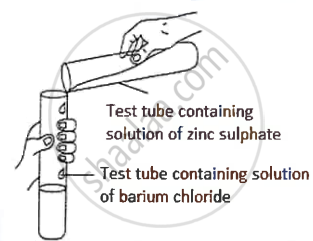
Write the word equation when ammonium salt is heated with caustic soda solution.
