Advertisements
Advertisements
प्रश्न
Calculate magnetic moment of thorium (Z=90). Is this element diamagnetic or paramagnetic?
Advertisements
उत्तर
The electronic configuration of thorium is [Rn] 5f0 6d2 7s2.
the condensed electronic configuration for its divalent cation will be
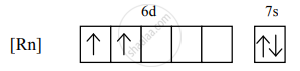
There are 2 unpaired electrons, so n = 2.
`therefore mu = sqrt(2 (2 + 2)) = sqrt8` BM = 2.83 BM
Spin only magnetic moment of the given ion is 2.83 BM. This element contains two unpaired electrons and hence, it is paramagnetic.
APPEARS IN
संबंधित प्रश्न
Choose the most correct option.
Magnetic moment of a metal complex is 5.9 B.M. Number of unpaired electrons in the complex is ______.
Answer the following
Why salts of Sc3⊕, Ti4⊕, V5⊕ are colourless?
Answer the following
Explain the trends in atomic radii of d block elements.
In 3d series, if nuclear charge increases, the shielding effects will ______.
Transition elements have more tendency to form interstitial compounds because _______.
Write formula to calculate magnetic moment.
Write any two uses of alloy.
Which of the following elements is alloyed with copper to form bronze?
Which ion has the highest value of theoretical magnetic moment?
Which hydride among the following is the strongest reducing agent?
The first element of 3d series is ____________.
The valence shell electronic configuration of V3+ ion is ____________.
Which of the following is not the mineral of iron?
The statement that is INCORRECT about the interstitial compounds is:
Among the following, ____________ has the largest atomic radius.
Which among the following substances is used as a catalyst for thermal decomposition of potassium chlorate?
Which of the following ions has d6 outer electronic configuration?
The following statement is CORRECT EXCEPT:
Which among the following pairs of elements in their respective oxidation states will have same value of effective magnetic moment?
(Atomic number: Sc = 21, Ti = 22, Cr = 24, Co = 27, Ni = 28, Zn = 30)
Identify the soft element from following.
What is the melting point of zinc?
What is the number of unpaired electrons of nickel observed in \[\ce{[Ni(CN)4]^{2-}}\]?
Which among the following compounds is not a colourless gas?
What two uses of the alloys.
Which element from the following does not exhibit a magnetic moment in the +1 state?
