Advertisements
Advertisements
प्रश्न
| Amines have a lone pair of electrons on nitrogen atom due to which they behave as Lewis base. Greater the value of Kb or smaller the value of pKb, stronger is the base. Amines are more basic than alcohols, ethers, esters, etc. The basic character of aliphatic amines should increase with the increase of alkyl substitution. But it does not occur in a regular manner as a secondary aliphatic amine is unexpectedly more basic than a tertiary amine in aqueous solutions. Aromatic amines are weaker bases than ammonia and aliphatic amines. Electron releasing groups such as −CH2, −OCH3, −NH2, etc., increase the basicity while electron-withdrawing substituents such as −NO2, −CN, halogens, etc., decrease the basicity of amines. The effect of these substitute is more at p− than at m− position. |
- Arrange the following in the increasing order of their basic character. Give reason: [2]
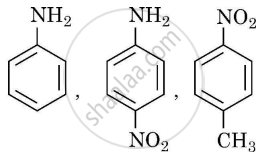
- Why pKb of aniline is more than that of methylamine? [1]
-
- Arrange the following in the increasing order of their basic character in an aqueous solution: [1]
(CH3)3N, (CH3)2NH, NH3, CH3NH2
OR - Why is ammonolysis of alkyl halides not a good method to prepare pure amines? [1]
- Arrange the following in the increasing order of their basic character in an aqueous solution: [1]
कारण सांगा
सविस्तर उत्तर
Advertisements
उत्तर
(a) The basicity of a compound increases by electron-donating groups, such as alkyl groups, while it decreases by electron-withdrawing groups, such as NO2 and CN. Therefore, the correct order of basicity of the given compounds can be determined accordingly.
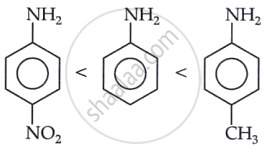
(b) In aniline, the lone pair of electrons on the N atom is delocalized into the benzene ring, reducing the electron density on nitrogen. As a result, aniline shows a weaker basic character compared to methylamine, and therefore, its pKb value is higher than that of methylamine.
(c)
- NH3 < (CH3)3N < CH3NH2 < (CH3)2NH
OR - This occurs because the primary amine produced can react with an alkyl halide to form a secondary amine. The secondary amine can then react again with the alkyl halide to produce a tertiary amine, which can undergo another reaction with the alkyl halide to form a quaternary ammonium salt.
shaalaa.com
या प्रश्नात किंवा उत्तरात काही त्रुटी आहे का?
2024-2025 (March) Outside Delhi Set 1
