Advertisements
Advertisements
Write the molecular formula and possible structural formula of the first four members of homologous series of carboxylic acids.
Concept: undefined >> undefined
Write the molecular formula of the first six members of homologous series of nitro – alkanes.
Concept: undefined >> undefined
Advertisements
Give example for the following type of organic reaction.
Electrophilic substitution
Concept: undefined >> undefined
\[\ce{C2H5Br + 2Na ->[dry ether]C4H10 + 2NaBr}\]
The above reaction is an example of which of the following.
Concept: undefined >> undefined
In the following reaction,
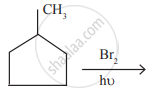
The major product obtained is
Concept: undefined >> undefined
Which of the following is optically active?
Concept: undefined >> undefined
The compounds formed at anode in the electrolysis of an aquous solution of potassium acetate are ______.
Concept: undefined >> undefined
The general formula for cyclo alkanes.
Concept: undefined >> undefined
Which of the following compounds shall not produce propene by reaction with HBr followed by elemination (or) only direct elimination reaction (NEET).
Concept: undefined >> undefined
Cis – 2- butene and trans – 2 – butene are ______.
Concept: undefined >> undefined
\[\begin{array}{cc}
\ce{CH2 - CH2 ->[(A)] CH ≡ CH}\\
|\phantom{.......}|\phantom{.................}\\
\ce{Br}\phantom{.....}\ce{Br}\phantom{.................}
\end{array}\], where A is,
Concept: undefined >> undefined
Describe the conformers of n – butane.
Concept: undefined >> undefined
Which of the following compounds will give racemic mixture on nucleophilic substitution by OH- ion?
- \[\begin{array}{cc}\ce{CH3 - CH - CH2Br}\\
|\phantom{....}\\\ce{C2H5}\end{array}\] - \[\begin{array}{cc}\ce{CH3}\\
|\phantom{.}\\\ce{H3C - C - C2H5}\\
|\phantom{.}\\\ce{Br}\end{array}\] - \[\begin{array}{cc}\ce{H}\\
|\phantom{.}\\\ce{CH3 - C - C2H5}\\
|\phantom{.}\\\ce{Cl}\end{array}\]
Concept: undefined >> undefined
The treatment of ethyl formate with excess of RMgX gives
Concept: undefined >> undefined
The carbocation formed in SN1 reaction of alkyl halide in the slow step is ______.
Concept: undefined >> undefined
The raw material for Raschig process
Concept: undefined >> undefined
Which alkyl halide from the following pair is
- chiral
- undergoes faster SN2 reaction?


Concept: undefined >> undefined
Complete the following reaction.
\[\ce{C6H5Cl + Mg ->[THF]}\]
Concept: undefined >> undefined
Complete the following reaction.
\[\ce{CHCl3 + HNO3 ->[\Delta]}\]
Concept: undefined >> undefined
An element X has the following isotopic composition 200X = 90%, 199X = 8% and 202X = 2%. The weighted average atomic mass of the element X is closest to
Concept: undefined >> undefined
