Advertisements
Advertisements
Choose the correct option from the given alternatives.
Which of the following is made up of polyamides?
Concept: undefined >> undefined
Answer the following in one sentence.
Identify 'A' in the following reaction:
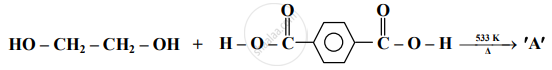
Concept: undefined >> undefined
Advertisements
Answer the following in one sentence.
Identify 'B' in the following reaction:
\[\ce{H2N -(CH2)6 - NH2 + HOOC - (CH2)4 - COOH ->[N2][533 K]}\]'B'
Concept: undefined >> undefined
Answer the following in one sentence.
Define the term: Vulcanization
Concept: undefined >> undefined
Answer the following in one sentence.
What type of intermolecular force leads to high-density polymer?
Concept: undefined >> undefined
Answer the following in one sentence.
Identify thermoplastic and thermosetting plastic from the following:
- PET
- Urea formaldehyde resin
- Polythene
- Phenol formaldehyde resin
Concept: undefined >> undefined
Answer the following.
Write the reaction of the formation of Nylon 6.
Concept: undefined >> undefined
Answer the following.
Write the reaction of the formation of Terylene.
Concept: undefined >> undefined
Answer the following.
Write structure of natural rubber and neoprene rubber along with the name and structure of their monomers.
Concept: undefined >> undefined
Answer the following.
Name the polymer type in which following linkage is present.
\[\begin{array}{cc}\ce{- C - O -}\\||\phantom{.....}\\
\ce{O\phantom{.....}}\end{array}\]
Concept: undefined >> undefined
Answer the following.
Match the following pairs:
| Name of polymer | Monomer |
| 1. Teflon | a. CH2 = CH2 |
| 2. PVC | b. CF2 = CF2 |
| 3. Polyester | c. CH2 = CHCl |
| 4. Polythene | d. C6H5OH and HCHO |
| 5. Bakelite | e. Dicarboxylic acid and polyhydoxyglycol |
Concept: undefined >> undefined
Draw the structures of polymers formed from the following monomers
\[\ce{n HOOC–R–COOH + n HO–R'–OH}\]
Concept: undefined >> undefined
Answer the following.
Draw the structures of polymers formed from the following monomers
H2N–(CH2)5 – COOH
Concept: undefined >> undefined
Name and draw structure of the repeating unit in natural rubber.
Concept: undefined >> undefined
Answer the following.
Write name and formula of raw material from which bakelite is made.
Concept: undefined >> undefined
Identify condensation polymers and addition polymers from the following.
\[\begin{array}{cc}\ce{-(CH2 - CH -)_{n}}\\
\phantom{....}|\\\ce{\phantom{.......}C6H5}
\end{array}\]
Concept: undefined >> undefined
Answer the following
1 nm = _________m ?
Concept: undefined >> undefined
Identify condensation polymers and addition polymers from the following.
-(CH2 - CH = CH - CH2 -)n
Concept: undefined >> undefined
Identify condensation polymers and addition polymers from the following.
-(CO(CH2)4 - CONH(CH2)6NH-)n
Concept: undefined >> undefined
Identify condensation polymers and addition polymers from the following.
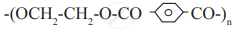
Concept: undefined >> undefined
