Please select a subject first
Advertisements
Advertisements
With the help of a diagram answer the questions are given below:
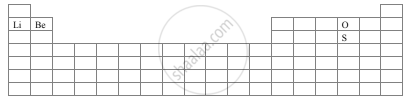
- Which atom should have smaller ionization energy, oxygen, or sulphur?
- The lithium forms +1 ions while beryllium forms +2 ions?
Concept: undefined >> undefined
Advertisements
Define electronegativity
Concept: undefined >> undefined
What are the valence electrons?
Concept: undefined >> undefined
Define ionization enthalpy.
Concept: undefined >> undefined
Name the factors on which ionization enthalpy depends?
Concept: undefined >> undefined
How does ionization enthalpy vary down the group and across a period?
Concept: undefined >> undefined
How the atomic size vary in a group and across a period? Explain with suitable example.
Concept: undefined >> undefined
Give reason.
Alkali metals have low ionization enthalpies.
Concept: undefined >> undefined
Give reason.
Inert gases have exceptionally high ionization enthalpies.
Concept: undefined >> undefined
Find out the type of isomerism exhibited by the following pair.
CH3 – CH2 – NH – CH2 - CH3 and CH3 - NH - CH2 - CH2 - CH3
Concept: undefined >> undefined
Find out the type of isomerism exhibited by the following pair.
\[\begin{array}{cc}
\ce{CH3 - CH - CH2 - CH3 and CH3 - CH2 - O - CH2 - CH3}\\|\phantom{...........................................}\\
\ce{OH}\phantom{.........................................}\end{array}\]
Concept: undefined >> undefined
Give reason.
Fluorine has less electron affinity than chlorine.
Concept: undefined >> undefined
Find out the type of isomerism exhibited by the following pair.

Concept: undefined >> undefined
Give reason.
Noble gases possess relatively large atomic size.
Concept: undefined >> undefined
Find out the type of isomerism exhibited by the following pair.

Concept: undefined >> undefined
Choose the correct option.
Which type of isomerism is possible in CH3 CHCHCH3?
Concept: undefined >> undefined
Answer in one sentence.
The pressure that each individual gas would exert if it were alone in the container, what do we call it as?
Concept: undefined >> undefined
Answer in one sentence.
When a gas is heated the particles move more quickly. What is the change in the volume of a heated gas if the pressure is kept constant?
Concept: undefined >> undefined
If the density of a gas is measured at constant temperature and pressure then which of the following statement is correct?
Concept: undefined >> undefined
