Please select a subject first
Advertisements
Advertisements
If 10−4 dm3 of water is introduced into a 1 dm3 flask at 300 K, how many moles of water are in the vapour phase when equilibrium is established?
(Given: Vapour pressure of H2O at 300 K is 3170 Pa; R = 8.314 JK−1 mol−1)
Concept: undefined >> undefined
The exothermic formation of ClF3 is represented by the equation:
\[\ce{Cl2(g) + 3F2(g) <=> 2ClF3(g)}\]; ΔH = −329 kJ
Which of the following will increase the quantity of ClF3 in an equilibrium mixture of Cl2, F2 and ClF3?
Concept: undefined >> undefined
Advertisements
The conversion of benzene diazonium chloride to bromobenzene can be accomplished by ______.
Concept: undefined >> undefined
The products obtained from a reaction of hydrogen peroxide and acidified potassium permanganate are ______.
Concept: undefined >> undefined
In a basic medium, H2O2 exhibits which of the following reactions?
(A) \[\ce{Mn^{2+} -> Mn^{4+}}\]
(B) \[\ce{I2 -> I^-}\]
(C) \[\ce{PbS -> PbSO4}\]
Choose the most appropriate answer from the options given below.
Concept: undefined >> undefined
The strength of the 11.2 volume solution of H2O2 is ______.
[Given that molar mass of H = 1 g mol−1 and O = 16 g mol−1)
Concept: undefined >> undefined
Insulin production and its action in human body are responsible for the level of diabetes. This compound belongs to which of the following categories?
Concept: undefined >> undefined
Which one of the following class of compounds is obtained by polymerization of acetylene?
Concept: undefined >> undefined
Which of the following order is correct:
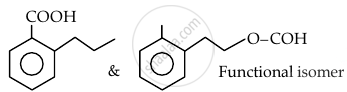
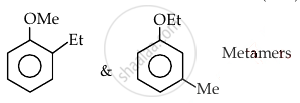
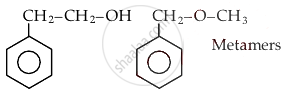
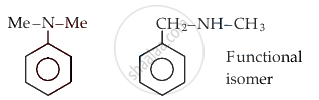
Concept: undefined >> undefined
Alkynes are isomers of ______.
Concept: undefined >> undefined
3.5 g of a fuel (with molecular weight 28), was burnt in a calorimeter and raised the temperature of 1 g water from 25° C to 67.3° C. If all the heat generated was used in heating water, the heat of combustion of fuel is ______ k cal.
Concept: undefined >> undefined
For micelle formation, which of the following statements are correct?
- Micelle formation is an exothermic process.
- Micelle formation is an endothermic process.
- The entropy change is positive.
- The entropy change is negative.
Concept: undefined >> undefined
20% surface sites have adsorbed N2. On heating N2 gas evolved from sites and were collected at 0.001 atm and 298 K in a container of volume is 2.46 cm3. Density of surface sites is 6.023 × 1014/cm2 and surface area is 1000 cm2, the no. of surface sites occupied per molecule of N2 is ______.
Concept: undefined >> undefined
Spin only magnetic moment of [MnBr6]4- is ______ B.M. (Round off to the closest integer)
Concept: undefined >> undefined
The spin-only magnetic moment value for the complex [Co(CN6)]4- is ______ BM. [At. no. of Co = 27]
Concept: undefined >> undefined
In an ore of iron, iron is present in two oxidation state Fen+ and `"Fe"^(("n"+1)+)`. Number of `"Fe"^(("n"+1)+)` is twice the number of Fen+. If empirical formula of ore is FexO. The value of [x × 100] is ______.
Concept: undefined >> undefined
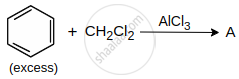
A is:
Concept: undefined >> undefined
A substance was known by its mode of synthesis to contain 10 atoms of carbon per molecule along with unknown number of atoms of chlorine hydrogen and oxygen. Analysis showed 60.5% carbon, 5.55% hydrogen, 16.10%j oxygen and 17.9% chlorine. The Empirical formula of the compound is ______.
Concept: undefined >> undefined
In a certain region of space there are only 5 molecules per cm3 on average. The temperature is 3 K. The average pressure of this very dilute gas is ______ × 10-21 atoms.
Concept: undefined >> undefined
The activating nature of \[\ce{- CH3}\] group linked to benzene ring can be explained with the help of ______.
Concept: undefined >> undefined
