Advertisements
Advertisements
प्रश्न
Spin only magnetic moment of [MnBr6]4- is ______ B.M. (Round off to the closest integer)
विकल्प
6 B.M.
3 B.M.
10 B.M.
8 B.M.
MCQ
रिक्त स्थान भरें
Advertisements
उत्तर
Spin only magnetic moment of [MnBr6]4- is 6 B.M.
Explanation:
[MnBr6]4-
x - 6 = - 4
x = + 2
Mn = [Ar] 3d5 4s2
Mn2+ = [Ar] 3d5 4s0
Mn2+ = [Ar] 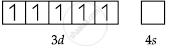
So, the number of unpaired electrons (n) = 5
`mu = sqrt("n" ("n + 2"))`
`mu = sqrt(5 (5 + 2))`
= `sqrt35`
= 5.91 B.M. ≈ 6 B.M.
shaalaa.com
Magnetic Moment of a Coil
क्या इस प्रश्न या उत्तर में कोई त्रुटि है?
