Advertisements
Advertisements
Why is hydrogen placed with alkali metals?
Concept: undefined >> undefined
Hydrochloric acid contains (i) hydrogen (ii) chlorine. Prove it. Write equations for the reactions.
Concept: undefined >> undefined
Advertisements
Name a black metallic oxide which reacts with hydrochloric acid to give a coloured solution.
Concept: undefined >> undefined
Name two gases which chemically combine to form a liquid.
Concept: undefined >> undefined
Name a chloride which is solube in excess of ammonium hydroxide
Concept: undefined >> undefined
Name the chemical in which gold can be dissolved.
Concept: undefined >> undefined
Name the experiment which demonstrates that hydrogen chloride is soluble in water.
Concept: undefined >> undefined
Give reason for the following:
silver nitrate solution can be used to distinguish HCl from HNO3
Concept: undefined >> undefined
Complete and balance the following reaction, state whether dilute or conc. acid is used.
\[\ce{NH4OH + HCl -> }\]
Concept: undefined >> undefined
Complete and balance the following reaction, state whether dilute or conc. acid is used.
\[\ce{NaHSO3 + HCl ->}\]
Concept: undefined >> undefined
Complete and balance the following reaction, state whether dilute or conc. acid is used.
\[\ce{Pb(NO3)2 + HCl ->}\]
Concept: undefined >> undefined
Complete and balance the following reaction, state whether dilute or conc. acid is used.
\[\ce{Pb3O4 + HCl ->}\]
Concept: undefined >> undefined
Complete the following reaction and balance them.
Zn+ 2HCl ⟶
Concept: undefined >> undefined
Take an element from an alkali metal and one from an alkaline earth metal and write an equation for their action with:
Hydrochloric acid,
Concept: undefined >> undefined
Complete the following reaction and balance them.
Ca(HCO3)2 + 2HCl →
Concept: undefined >> undefined
Take an element from an alkali metal and one from an alkaline earth metal and write an equation for their action with:
Oxygen
Concept: undefined >> undefined
Take an element from an alkali metal and one from an alkaline earth metal and write an equation for their action with:
Sulphuric acid
Concept: undefined >> undefined
Take an element from an alkali metal and one from an alkaline earth metal and write an equation for their action with:
Water.
Concept: undefined >> undefined
Study the flow chart and give balanced equations with conditions for the conversions A, B, C, D and E.
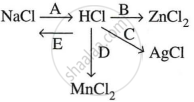
Concept: undefined >> undefined
Write the balanced equation for the reaction between ammonia and sulphuric acid.
Concept: undefined >> undefined
