Advertisements
Advertisements
For the reaction
`2AgCl (s) + H_2 (g) ("1 atm") -> 2Ag (s) + 2H^(+) (0.1 M) + 2Cl^(-) (0.1 M)`
`triangleG^0 = -43600 J at 25^@ C`
Calculate the e.m.f. of the cell
`[log 10^(-n) = -n]`
Concept: undefined >> undefined
Draw the structures of the following :
H2S2O7
Concept: undefined >> undefined
Advertisements
Which of the two components of starch is water soluble?
Concept: undefined >> undefined
Define the following with a suitable example, of each:
Coagulation
Concept: undefined >> undefined
What is observed when an emulsion is centrifuged?
Concept: undefined >> undefined
Out of starch and ferric hydroxide sol, which one can easily be coagulated and why?
Concept: undefined >> undefined
Write equations of the following reactions:
Acetylation of aniline
Concept: undefined >> undefined
An unripe mango placed in a concentrated salt solution to prepare pickle, shrivels because ______.
Concept: undefined >> undefined
Consider the figure and mark the correct option.
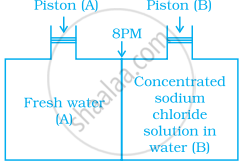
Concept: undefined >> undefined
Water retention or puffiness due to high salt intake occurs due to ______.
Concept: undefined >> undefined
Why are aquatic species more comfortable in cold water in comparison to warm water?
Concept: undefined >> undefined
When kept in water, raisin swells in size. Name and explain the phenomenon involved with the help of a diagram. Give three applications of the phenomenon.
Concept: undefined >> undefined
In the preparation of \[\ce{H2SO4}\] by Contact Process, why is \[\ce{SO3}\] not absorbed directly in water to form \[\ce{H2SO4}\]?
Concept: undefined >> undefined
Write the structure of pyrophosphoric acid.
Concept: undefined >> undefined
Match the formulas of oxides given in Column I with the type of oxide given in Column II and mark the correct option.
| Column I | Column II |
| (A) \[\ce{Pb3O4}\] | (1) Neutral oxide |
| (B) \[\ce{N2O}\] | (2) Acidic oxide |
| (C) \[\ce{Mn2O7}\] | (3) Basic oxide |
| (D) \[\ce{Bi2O3}\] | (4) Mixed oxide |
Concept: undefined >> undefined
Assertion: \[\ce{SF6}\] cannot be hydrolysed but \[\ce{SF4}\] can be.
Reason: Six \[\ce{F}\] atoms in \[\ce{SF6}\] prevent the attack of \[\ce{H2O}\] on sulphur atom of \[\ce{SF6}\].
Concept: undefined >> undefined
\[\begin{array}{cc}
\ce{CH3 - C ≡ CH ->[40{%} H2SO4][1{%} HgSO4] A ->[Isomerisation] CH3 - C - CH3}\\
\phantom{........................................}||\\
\phantom{........................................}\ce{O}\\
\end{array}\]
Structure of ‘A’ and type of isomerism in the above reaction are respectively.
Concept: undefined >> undefined
Arenium ion involved in the bromination of aniline is:
(i)

(ii)

(iii)

(iv)

Concept: undefined >> undefined
Why is benzene diazonium chloride not stored and is used immediately after its preparation?
Concept: undefined >> undefined
Why does acetylation of –NH2 group of aniline reduce its activating effect?
Concept: undefined >> undefined
