Advertisements
Advertisements
प्रश्न
For the reaction
`2AgCl (s) + H_2 (g) ("1 atm") -> 2Ag (s) + 2H^(+) (0.1 M) + 2Cl^(-) (0.1 M)`
`triangleG^0 = -43600 J at 25^@ C`
Calculate the e.m.f. of the cell
`[log 10^(-n) = -n]`
Advertisements
उत्तर
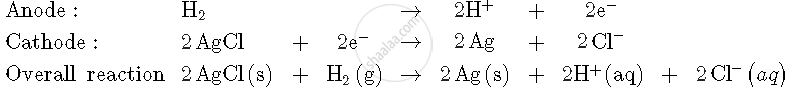
`triangleG^0 = -nFE_(cell)^0`
`:. E_(cell)^0 = 0.226 V`
`E_(cell) = 0.226 - (0.059)/2 log (0.1)^2 (0.1)^2`
Therefore, `E_(cell) = 0.226 - (0.059)/2 log 10^(-4)`
= 0.226 + 0.059 x 2
= 0.344 V
shaalaa.com
क्या इस प्रश्न या उत्तर में कोई त्रुटि है?
