Advertisements
Advertisements
Predict the product of the following reaction:
\[\ce{(CH3)3C - OC2H5 ->[HI]}\]
Concept: undefined >> undefined
Show how will you synthesize cyclohexylmethanol using an alkyl halide by an SN2 reaction.
Concept: undefined >> undefined
Advertisements
Write the equation of the reaction of hydrogen iodide with 1-propoxypropane.
Concept: undefined >> undefined
Write the equation of the reaction of hydrogen iodide with methoxybenzene.
Concept: undefined >> undefined
Write the equation of the reaction of hydrogen iodide with benzyl ethyl ether.
Concept: undefined >> undefined
Explain the fact that in aryl alkyl ethers
- the alkoxy group activates the benzene ring towards electrophilic substitution and
- it directs the incoming substituents to ortho and para positions in the benzene ring.
Concept: undefined >> undefined
Write the mechanism of the reaction of HI with methoxymethane.
Concept: undefined >> undefined
Why does O3 act as a powerful oxidising agent?
Concept: undefined >> undefined
How is O3 estimated quantitatively?
Concept: undefined >> undefined
Which aerosols deplete ozone?
Concept: undefined >> undefined
Write the product(s) in the following reaction
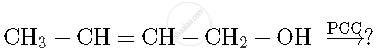
Concept: undefined >> undefined
Write the formula of reagents used in the following reactions :
Bromination of phenol to 2,4,6-tribromophenol
Concept: undefined >> undefined
Write the formula of reagents used in the following reactions :
Hydroboration of propene and then oxidation to propanol.
Concept: undefined >> undefined
What happens when CdCl2 is doped with AgCl?
Concept: undefined >> undefined
Calculate the freezing point of a solution containing 60 g of glucose (Molar mass = 180 g mol–1) in 250 g of water. (Kf of water = 1.86 K kg mol–1)
Concept: undefined >> undefined
Account for the following:
pKa value of 4-nitrobenzoic acid is lower than that of benzoic acid.
Concept: undefined >> undefined
Give reasons for the following:
Measurement of osmotic pressure method is preferred for the determination of molar masses of macromolecules such as proteins and polymers.
Concept: undefined >> undefined
(A), (B) and (C) are three non-cyclic functional isomers of a carbonyl compound with molecular formula C4H8O. Isomers (A) and (C) give positive Tollens' test whereas isomer (B) does not give Tollens' test but gives positive Iodoform test. Isomers (A) and (B) on reduction with Zn(Hg)/conc. HCl give the same product (D).
1) Write the structures of (A), (B), (C) and (D).
2) Out of (A), (B), and (C) isomers, which one is least reactive towards the addition of HCN?
Concept: undefined >> undefined
Write the structures of the main products in the following reactions :

Concept: undefined >> undefined
Define the following with an example
Denatured protein
Concept: undefined >> undefined
